Modality Review: Introduction
to Ultrasound for Preclinical Imaging
When to Consider Using Ultrasound
Ultrasound (US) imaging is one of the most commonly used diagnostic techniques clinically, and is widely used in preclinical imaging as well. It is a safe, non-invasive, and relatively inexpensive technology compared to some of the other available imaging modalities. As it does not emit or require the use of ionizing radiation, it is well suited for use in preclinical longitudinal studies. With the development of higher frequency transducers, increased resolution is accomplished, allowing the smaller structures of a variety of preclinical disease models to be imaged.
Ultrasound imaging is ideally suited for imaging a variety of soft tissues, including the heart, blood vessels, abdominal organs, urogenital tract, muscles and tendons etc. The main limitations of ultrasound imaging, is that the sound waves used to generate the images cannot penetrate calcified bone, or air very well; making the lungs, brain, and bone marrow challenging to image without some adaptations within the model at the time of imaging.
Typical ultrasound systems utilize a variety of imaging modes to examine different qualities of the imaging subject. For example, B-mode (brightness mode) is used to generate 2-dimensional greyscale images, and are typically displayed in real time (30 to 130 frames per second). While M-mode (motion mode) images are acquired along a single line, and typically show movement of the myocardium or blood vessel walls. Doppler techniques are used to visualize and quantify blood flow in various vessels throughout the body and within the heart. Color and Power Doppler techniques provide a color overlay of where blood is flowing within a variety of organs and blood vessels; while Pulsed Wave Doppler is used to quantify the velocity at which the blood is flowing through various blood vessels. Finally, Tissue Doppler allows users to assess the movement of tissue, for example at the annulus of the mitral/tricuspid valves, where changes in tissue movement often precede changes in blood flow. Additional capabilities exist with some ultrasound systems, including the addition of a motor which translates the transducer over an area of interest to generate a 3-dimensional image. Image guided injections may also be performed, as both steel and glass needles are easily visualized on a B-mode ultrasound image. Finally, there are some advanced techniques, such as shear wave elastography, analysis of the native digital RF data, and programming of a variety of pulse sequences which may be explored by a variety of users such as those more interested in medical biophysics.
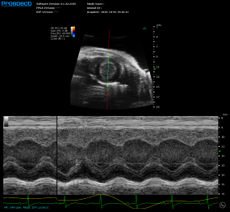
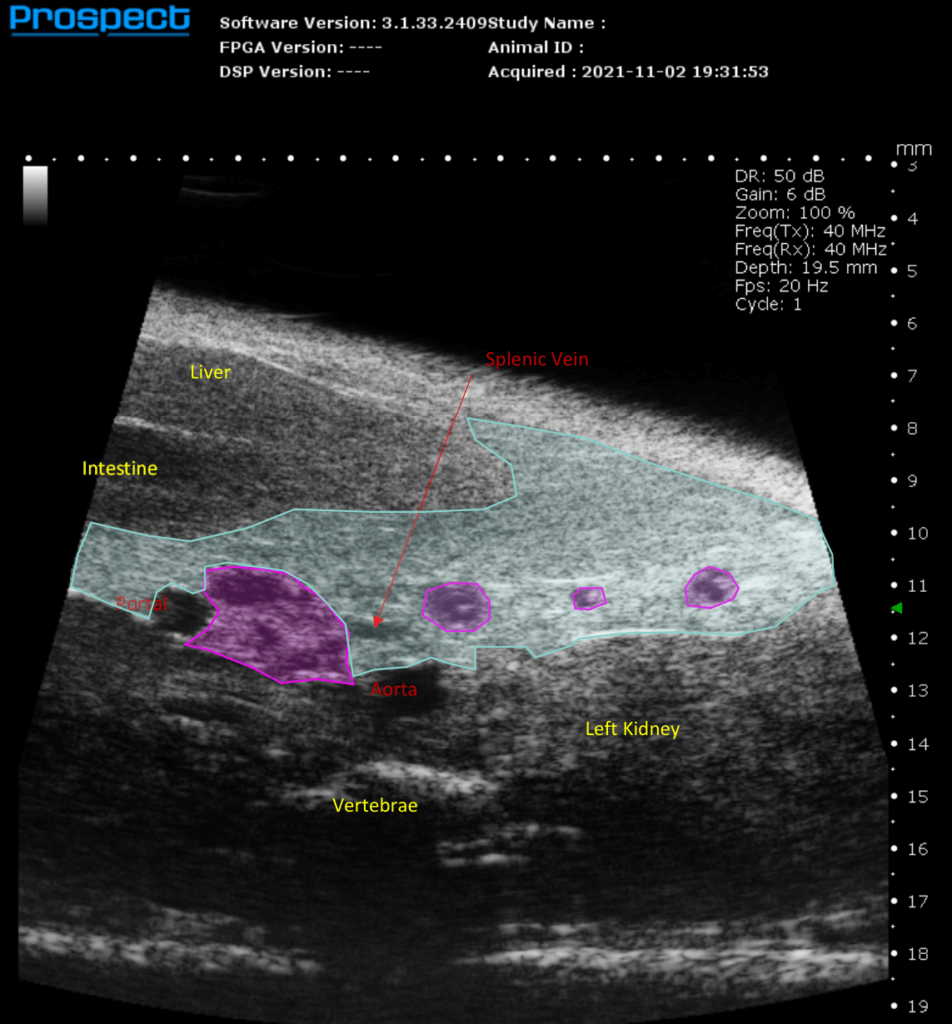
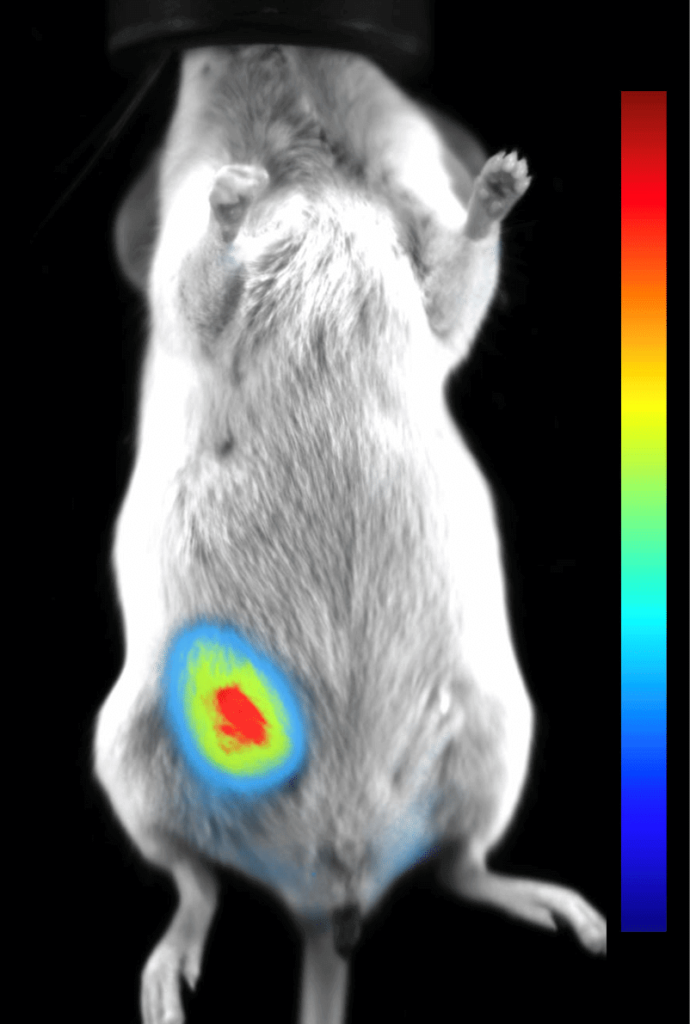
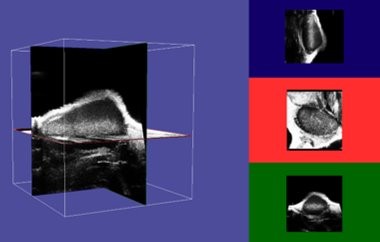
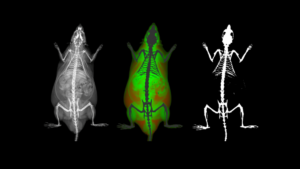
Multimodal Imaging
When considering the simplest meaning of multimodal imaging – that is to obtain data of multiple types from the same imaging subject, then an ultrasound system alone can do this. That is to obtain anatomical information about the heart using a B-Mode image, while also being able to assess the functional aspects of the heart as well using PW or Color Doppler. One may also consider expanding this capability by using a microbubble contrast agent to either look at perfusion within a specific organ, or, in the case of a targeted microbubble, to look at the expression of a specific biomarker on the endothelial cell surface, i.e. expression of VEGF-R2 in angiogenic vessels within a tumor.
However, expanding the definition of multimodal imaging to encompass multiple imaging modalities is also possible using ultrasound imaging. In many studies ultrasound is incorporated to visualize a variety of soft tissues or disease pathologies, which may be indicated by another modality. For example, bioluminescence (BLI) or fluorescence (FLI) may be used to provide an indication of where a metastatic tumor is located within the whole body of an animal model. While ultrasound can then be used to visualize the tumor, confirm its location, and quantify its volume using 3D techniques. Likewise, in a toxicology study, the target compound may be tagged with a fluorophore or a radionucleotide (PET or SPECT) and one may use one of these technologies to examine the biodistribution of the compound within the whole body, but then use ultrasound to study the effect that compound has on cardiac function, for example.
The complimentary nature of all imaging modalities, including ultrasound, is such that the capabilities of each within a specific study is only limited by access to the equipment, and thinking or considering the possible outcomes that are available.
High Frequency Ultrasound System – Accessible in Everyone
When considering an ultrasound system for preclinical imaging it is important to focus on those systems specifically designed for use on small animal models. Of upmost importance is to work with the highest frequency sound possible, to achieve the best resolution to resolve the small anatomical structures in these models, while maintaining the required depth of penetration needed to reach the imaging target. However, there are other considerations as well – including the integration of anesthesia, if required, maintaining and monitoring normal physiological parameters, and ensuring the reproducible nature of the data.
For most, the use of an animal handling station is desirable for a few reasons. Most will have a table or platform on which the animal will be positioned. This has many functions, but should be heated, to help maintain the temperature of the animals, and should provide monitoring of the heart rate, respiration rate, and body temperature. Additionally, this table will help the user to achieve the correct positioning of the animal, in terms of the angles and stability needed. Additionally, the station will have a component to hold the ultrasound transducer; this will allow for stable and reproducible imaging.
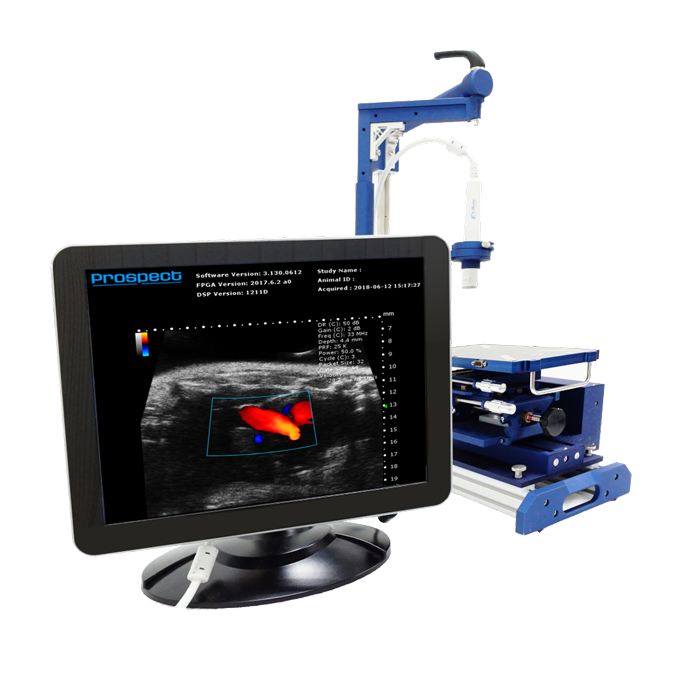
The Prospect T1 high frequency ultrasound system is manufactured by S-Sharp, and distributed by Scintica. This system is well-equipped and designed specifically for use on small animals such as mice, rats, and similarly sized animals. This system can be configured with probes ranging from 15-70MHz, allowing for resolution as low as 30µm. There are two available platforms, one sized for mice and the other for rats, which are easily interchangeable. These platforms are heated, have integrated anesthesia, and monitor heart rate, ECG, respiration rate, and body temperature. The animal handling station provides all axes of movement and flexibility which is required to get even the most challenging view required for cardiac imaging. The probe holder is also highly flexible, providing all movements and angles that are required.
The Prospect T1 is configured with all available imaging modes, including access to the raw digital RF data, which some users may wish to analyze for more advanced findings. There are three available options (1) 3D motor for organ and tumor volumes, (2) image guided injection mount, (3) shear wave elastography.
The Prospect T1 is a tablet-based system, with all of the electronics sitting behind the screen, reducing the footprint of this system dramatically compared to most other commonly used systems in preclinical imaging. This system performs very well for most of the common imaging applications including cardiac function, blood vessel imaging, phenotyping, cancer research, developmental biology, ophthalmology, etc. and also includes many advanced features like access to the raw data, and ability to perform shear wave elastography. The Prospect T1 is priced to allow most researchers direct access to the system within their own lab, without the need to access a core imaging lab system which may come with its own challenges such as user fees, technician fees, difficult scheduling hours, and the need to transport animals.
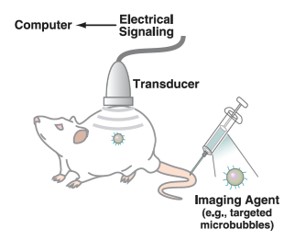
Understanding the Basics of Ultrasound Imaging
At its most basic level, ultrasound imaging can be considered as sound in and sound out to create an image. The soundwaves used in ultrasound imaging are generated by piezoelectric elements/crystals within the probe/transducer used for imaging. These piezoelectric crystals are typically made up of quartz or ceramic materials, elastic vibrations are created within the crystals which ultimately propagate soundwaves into the material being investigated – called the piezoelectric effect. This effect is created when the crystals are charged with alternating electrical voltage. The soundwaves are then reflected off various objects, in the example of small animal imaging, off various tissues within the imaging subject. The reflected soundwaves are then also received by the piezoelectric crystals which convert them into electrical signals, and the electronics within the system interpret these into images displayed by the system.
The reflected portion of the transmitted soundwave depends heavily on the materials it interacts with, and that which it must pass through. Additionally, the angle at which the soundwave encounters the material will affect the reflection of the wave as well. This becomes important when considering the visualization of a specific structure in B-mode imaging. The strongest reflection of the soundwaves will occur when, for example, a blood vessel wall, is images directly perpendicular to the path of the soundwaves, to ensure the strongest reflection, and therefore best image. Additionally, soundwaves do not travel through air as efficiently as aqueous or water-based substances, such as ultrasound gel. Thus, ultrasound gel is important to act as an interface between the transducer and the imaging subject to ensure as much of the soundwave is transmitted to the imaging subject, and not absorbed into the air, which would weaken the resulting signal.
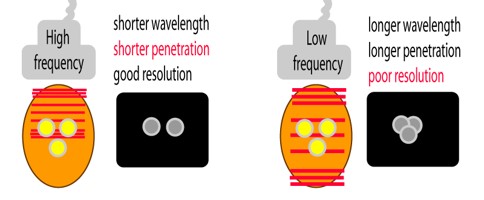
Understanding the principles of soundwaves is important to understanding how ultrasound works, and an image is created. However, there are many different wavelengths, or frequencies, of sound that one could choose from. Lower frequency sound penetrates tissues and media well, and is not as susceptible to absorption in air, as higher frequencies, so at first glance would appear to be an ideal choice. However, the image resolution is directly correlated to the frequency of the soundwaves. Thus, for small animal imaging, where target organs or structures are quite small, then higher frequency soundwaves should be used. One must consider, however, the depth of penetration that is required to reach the target of interest and adjust the frequency of sound accordingly.
When creating a 2-dimensional B-mode image, the system electronics receive the reflected soundwaves and consider the returning amplitude of the wave and interpret that into various levels of greyscale. The higher the amplitude of the returned wave, the brighter the greyscale value. In this way the system is able to look over many lines that make up the B-Mode image. When wishing to look along a single line, at a specific anatomical target (i.e., blood vessel wall, or myocardium) the user may locate the M-Mode sample volume in a specific location. Here the ultrasound system will display the movement of the tissue along that single line over time. In both B-Mode and M-Mode, the ultrasound system detects the amplitude of the reflected soundwave, to determine the greyscale intensity of the image, and looking at how that changes over time.
Expanding B-mode capabilities from 2-dimenstions to 3, increased the utility of an ultrasound system to allow for volume calculations of many targets, for example specific organs, focal lesions within those organs including cysts, tumor, etc. This is accomplished on most preclinical imaging systems by attaching a programable motor to either the probe, or the animal handling platform. In either case, the user defines how far the motor should travel, and the gap between each acquired 2D B-mode image. The software then stacks the multiple 2D images together generating a 3D image. The user may then define the edges of the area of interest, and the system calculates the defined volume.
Ultrasound imaging can also be used to assess blood flow, within a variety of blood vessels and within the chambers of the heart. The Doppler effect is key here – it states that frequency changes occur in the soundwaves if transmitters (i.e., piezoelectric crystals within the transducers) and receivers/reflectors (i.e., red blood cells within the blood vessels) move in relation to one another. A real-life example of this is the sound emitted by a firetruck as it moves towards a pedestrian; the frequency of the sound increases as the firetruck moves towards the pedestrian, while it decreases as the firetruck moves away from the pedestrian. When thinking about the Doppler effect with respect to blood flow, the ultrasound system transmits a known frequency of sound from the transducer, however the soundwave reflected off of a moving object, like a red blood cell, will be received at a different frequency. The ultrasound system must then determine the change in frequency of the soundwaves, thus providing an indication of both the velocity and direction that the blood cell was moving with. As one can imagine, the angle between the transmitted soundwave, and the movement of the red blood cell, for example, is of upmost importance to ensure the reflected soundwave returns to the transducer with the highest intensity. This angle becomes very important when working to quantify blood flow velocity over time, as is done with Pulsed Wave Doppler.
The Doppler effect may be used to create a color flow map which is overlaid on a 2-dimensional B-mode image. Conventions exist, when displaying the color flow map in Color Doppler Mode, to display blood moving Away from the transducer in Blue, while blood moving Towards the transducer is Red. In the case where direction of blood flow is not relevant, for example in tumor imaging, Power Doppler may be used, in which no directional information is provided, the presence of blood is shown in shades of orange typically, with tones closer to white being of higher velocity then darker tones. If one wishes to examine the blood flow velocity within a specified sample volume, (i.e., within the inflow jet of blood flowing between the left atrium and the left ventricle, through the mitral valve, of the heart) then soundwaves only from this region are examined, and the resulting blood flow velocity is displayed over time, with directional information included.
When discussing blood flow, as detected using the various Doppler techniques available, one is examining flow through large and smaller blood vessels, however this technique cannot be used to assess flow through the capillaries, for example. Flow in these small vessels, or perfusion within an organ or tumor, must be assessed using a contrast agent. Ultrasound contrast agents have traditionally been gas filled microbubbles, that is a bubble having a biocompatible outer shell (i.e., phospholipid) and a biocompatible gaseous core (i.e., perfluorocarbon gas). Due to their unique physical and acoustic characteristics the microbubbles can be imaged using ultrasound without destruction. When soundwaves of a low intensity/power strike a microbubble the rhythm of the acoustic wave causes the microbubbles to expand symmetrically through the negative and positive pressure waves created. In doing so, the microbubbles create an acoustic wave, of the same frequency of sound, which is received by the transducer, and can be converted into an image. When soundwaves or greater amplitude are used, the same phenomena occurs where the microbubbles expand and contract, however in this instance the microbubbles send back not only the same frequency of sound, but additional harmonic and subharmonic tones to that of the fundamental (or initial) frequency. Normal tissues on the other hand, do not create these harmonic and subharmonic tones.
When considering microbubble imaging, there are two common approaches – harmonic imaging, and pulse inversion imaging. In harmonic imaging, filters are used to isolate the harmonic signal coming from the microbubbles, which is not emitted from the surrounding tissue. This, however, has some drawbacks including reduced spatial resolution and less contrast. However, if pulse inversion is used these drawbacks may be avoided. In pulse inversion, two sequential pulses are sent, one inverted with respect to the other. Tissue signals, which come back at the same frequency, will cancel each other out, while signal from microbubbles will not, resulting in a highly sensitive contrast agent imaging technique.
One final technique, which some more advanced users are interested in is called shear wave elastography. In this technique, a secondary piezoelectric crystal is mounted onto the imaging probe, or specific crystals are programmed to create the additional soundwave. A non-imaging soundwave is sent into the tissue, and the reflected waves are received by the same crystal. The ultrasound system then determines the speed of sound through the specified 2-dimensional area of the tissue. A graphical representation of the calculated speeds is created, and the user may define specific regions of interest to quantify the speeds. The speed of sound through a specific tissue is dependent on the make-up of that tissue, down to the microscopic level. Elastography may be used to detect changes in tissue composition prior to changes in image characteristics that could be visualized. These tissue changes may be global changes, as may be seen in organ wide fibrosis, or accumulation of fat, or focal/regional changes.
Our Next Modules
Preclinical Imaging Modalities
Available Now
As we begin to explore the idea of multimodal imaging, let’s first start to explore the variety of preclinical imaging modalities that are most commonly used by researchers around the world.
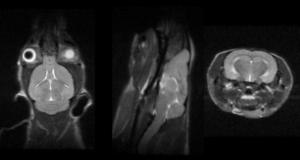
Modality: MRI
Available Now
MRI is considered the gold standard in soft tissue imaging, both in the clinic on patients and by researchers on a wide variety of preclinical imaging subjects.
Modality: CT
Available Now
Computed Tomography (CT) is one of the most commonly used clinical imaging techniques, next to perhaps ultrasound.
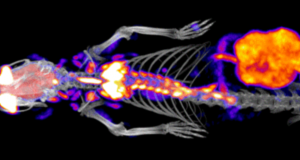
Modality: PET
Available Now
Nuclear imaging modalities include positron emission tomography (PET) as well as single photon emission computed tomography (SPECT), which both detect gamma radiation emitted from a radionucleotide injected into an imaging subject.
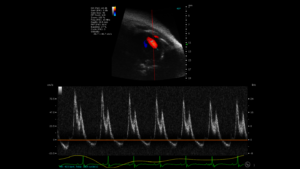
Modality: Ultrasound
Available Now
Ultrasound (US) imaging is one of the most commonly used diagnostic techniques clinically, and is widely used in preclinical imaging as well. It is a safe, non-invasive, and relatively inexpensive technology compared to some of the other available imaging modalities.
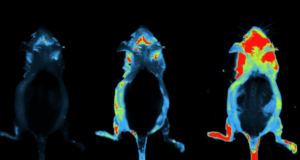
Modality: Optical
Available Now
When discussing Optical Imaging techniques in this series, the meaning is to cover bioluminescence (BLI) and fluorescence (FLI) imaging. T
Modality: Photoacoustic
Available Soon
Photoacoustic (PAI) imaging is a technology which combines the sensitivity of optical imaging with the depth of penetration and resolution of ultrasound imaging.
Modality: Microscopy
Available Soon