Modality Review: Introduction to CT for Preclinical Imaging
When to Consider Using CT
Computed Tomography (CT) is one of the most commonly used clinical imaging techniques, next to perhaps ultrasound. With recent advancements in scanners not only do CT images provide exquisite details of bone and air-filled structures such as the lungs, but further advancements have allowed for a level of soft tissue imaging as well. These clinical CT systems, like other clinical imaging systems, have been grossly adapted to work effectively with small animal imaging subjects. There have had to be many adaptations specific to CT, to help ensure sufficient image resolution, with appropriate radiation dose levels to allow for in vivo longitudinal studies.
When using the term micro (µ) CT, it is of upmost importance to be clear on what is meant. For some µCT simply refers to the adaptation of CT to work with small animal models, while for others µCT implies very high-resolution imaging which is carried out on ex vivo samples due to the high radiation doses required. Systems are most often designed for in vivo or ex vivo imaging, and not usually both, as there are significant differences in functional requirements and the best ways to achieve those in the most economical way.
Preclinical in vivo CT images are typically taken of the whole body to provide anatomical imaging mostly of the skeletal structures; however, some soft tissue contrast is possible. CT imaging is relevant for those studying cancer biology, neurology, specific organs such as the kidney, liver, but is one of the best imaging modalities to examine the lungs.
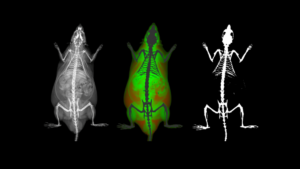
Multimodal Imaging
When considering multimodal imaging applications CT is again one of the most commonly used modalities to provide in vivo anatomical context to other imaging modalities, such as PET and SPECT, which do not provide the end user with sufficient anatomical detail.
In many cases, preclinical imaging systems include a CT module in which the initial image is acquired, followed sequentially by a CT acquisition. In these situations, the animal bed generally moves from one modality to the other, and the images are co-registered afterwards.
When a study reaches the end point, high resolution CT images may also be acquired to provide additional details about a specific pathology, most often used in bone metabolic research. These results, when combined with other imaging results provide further information to help elucidate the specific biological mechanisms at play in the disease model.
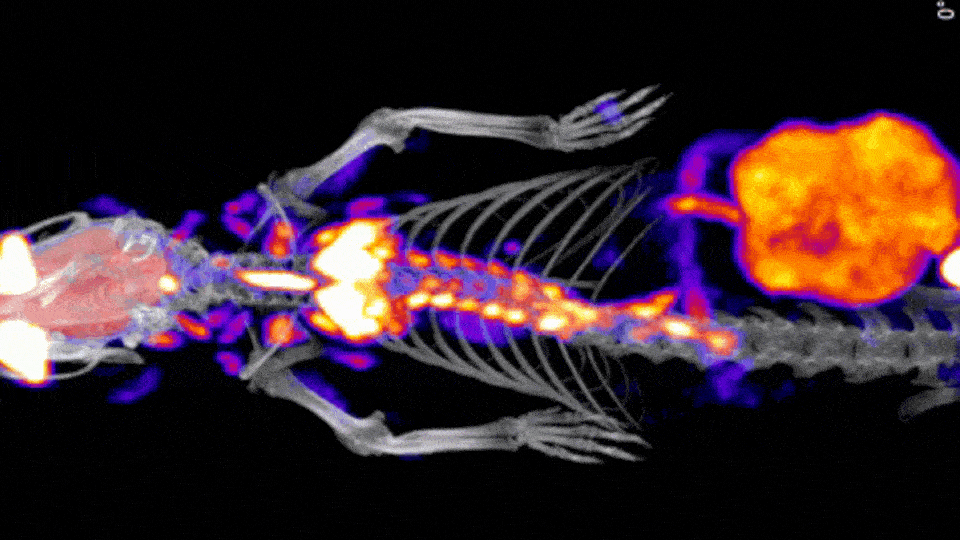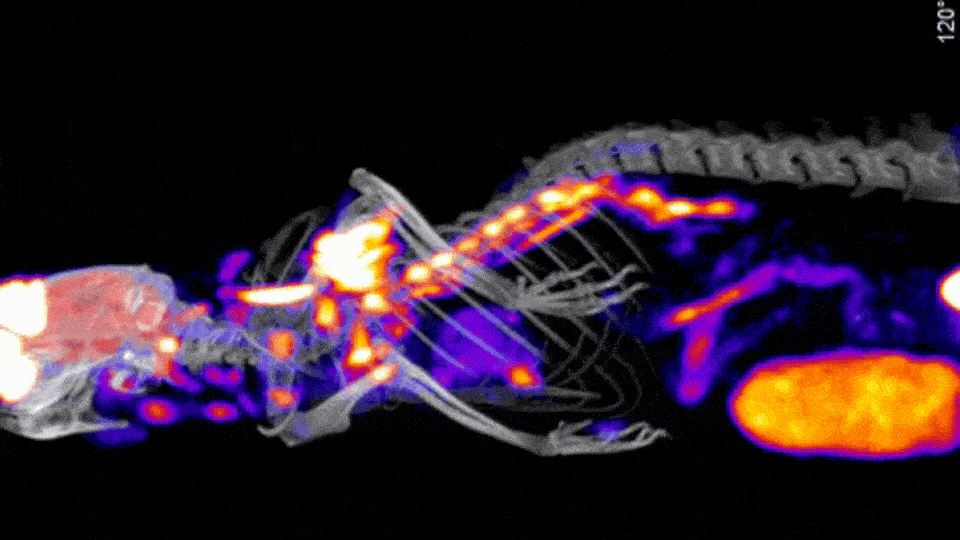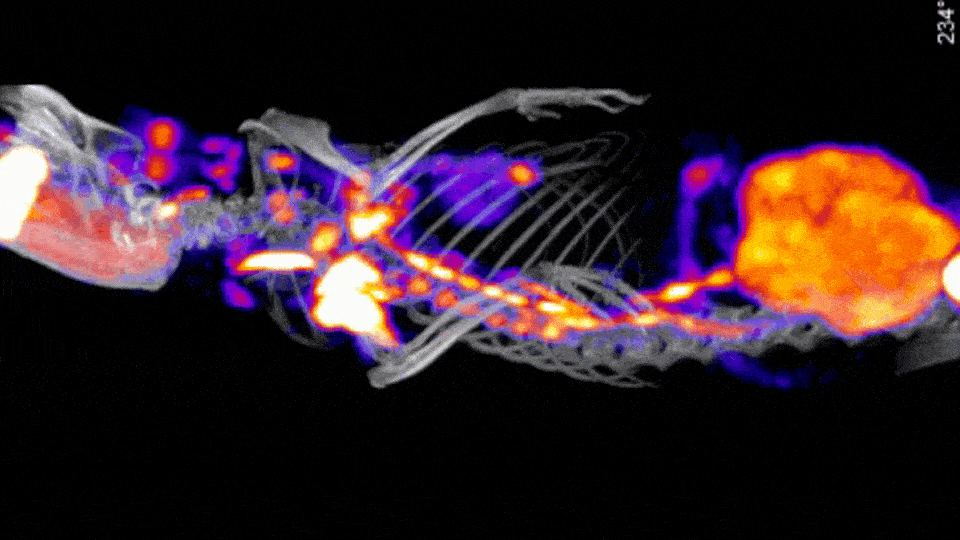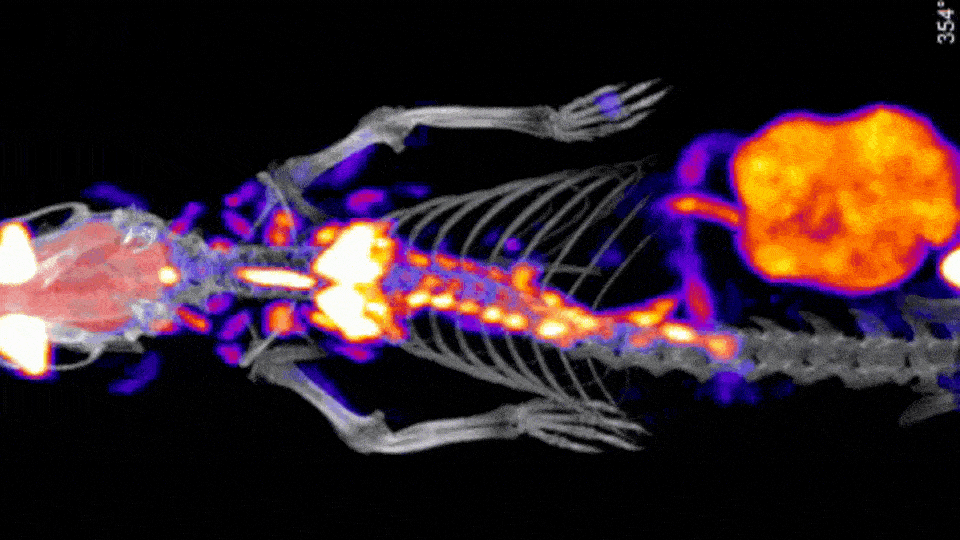

Stand Alone and Sequential In Vitro CT Solutions
There are a few CT options offered by Scintica, including a Compact CT designed for in vivo mouse-only imaging, along with the SuperArgus Basic and Advanced CT options which can operate as a stand-alone option or as part of a sequential PET/CT system. All of these systems provide rapid image acquisition, with 50-200µm image resolution, using radiation doses appropriate for in vivo longitudinal studies.
The Compact CT has been designed for mouse-only applications and the animal bed can easily be moved from the CT to the Compact PET system for multimodal imaging. Likewise, a simplistic multimodal imaging cassette can be used to move the animal from the Compact CT into other imaging modalities such as the Newton Bioluminescence/Fluorescence imaging system, or the M-Series compact MRI system.
The SuperArgus Basic and Advanced CT options are designed with a 70mm and 120mm transaxial field of view respectively. These systems have been designed for mice, rats, rabbits and similarly sized imaging subjects, with the ability to do multi-animal imaging as well. These systems can be added onto the SuperArgus PET systems, to perform sequential PET/CT imaging, or can be used by themselves, again allowing the animals to be moved to other imaging modalities as desired.
All CT systems can include integrated anesthesia, animal heating, as well as physiological monitoring. Finally, all systems are self-shielded, making them easy to install within an existing laboratory space, or within an animal facility.
Understanding the Basics of Computed Tomography
The idea of a 3-dimensional reconstructed image comes from the issues presented within a 2-dimensional x-ray image. That is that all of the tissue, bone, and structures located between the x-ray source, and the detector are displayed together; even when perpendicular images are taken, it is still challenging for one to reconstruct the position of all of the structures in 3-dimensional space. However, when a high number of 2D projections are acquired, a 3D images may be reconstructed and re-sampled as needed.
When a CT image is acquired, the x-ray source and detector are typically rotated around the imaging subject. This can be done in a step and shoot fashion, where a full 360o rotation is completed for each z-axis length. If additional length along the z-axis is required, another image is taken and combined with the previous scan. Alternatively, but less common in preclinical imaging, is to use a spiral trajectory, where the image is acquired while the imaging subject is translated along the z-direction. This spiral technique is used commonly in the clinic but is less favorable in preclinical imaging as the imaging subject may be exposed to a higher dose of radiation.
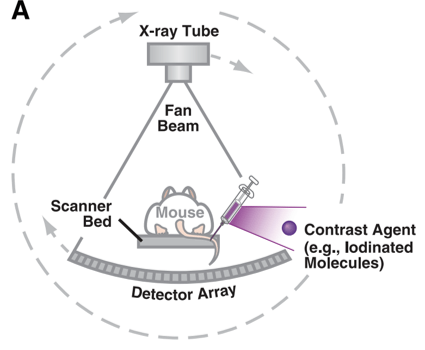
Figure adapted from – James ML, Gambhir SS. Physiological reviews. 2012
In order to perform the 3D image reconstruction, one must understand that each of the thousands of measurements taken in a single rotation of the gantry must be evaluated and used to help in the reconstruction. Of importance are the physical characteristics of the imaging subject – it’s thickness, attenuation coefficients, etc. When considering a small animal model for example, one must also recognize that the attenuation coefficients of the overlapping tissues, such as lungs, ribs, heart, muscle, and skin, must all be considered and distinguished in the reconstruction. There are many different approaches to image reconstruction, which may be considered the reconstruction kernel, the selection of which can affect the image sharpness/resolution and noise characteristics. One example of a reconstruction type is the filtered back project (FBP), while most current preclinical systems use a Feldkamp type reconstruction.
When images are displayed, they are most often displayed using the standard of Hounsfield units (HU), which can be thought of density relative to the density of water; where air has a value of -1000 HU, and water has a value of 0 HU. So, if a measured tissue has a value of 200 HU, then it can be thought of to have a density that is 1.2 times that of water. Typical HU values range from -1000 to +4000 HU. When an image is displayed, it typically is shown in greyscale; the user may adjust the window and level to determine the appearance of the image.
There are a few considerations when determining how best to perform CT imaging – that is whether it should be done in vivo in a longitudinal study, or ex vivo or at the end of a study. The main consideration is the radiation dose required to achieve the desirable resolution. When using CT imaging within an in vivo longitudinal study, the radiation dose must be minimized, so as to not interfere with the progression or the study. The required dose will depend on what is being visualized; if low-contrast imaging is desired to visualize more details within the soft tissues, then the dose must be optimized. However, if the image is acquired at the end point of a study, or on ex vivo samples, then higher doses of radiation may be used, and higher resolutions images acquired.
Dose and image quality are inherently related – high spatial resolution demands higher radiation dose, unless higher noise can be accepted. While if low radiation dose is required, this will increase noise, making low contrast imaging more challenging. There are significant efforts to reduce image noise, to allow for higher image quality with lower dose and scan times, which is ideal for in vivo longitudinal studies.
Our Next Modules
Preclinical Imaging Modalities
Available Now
As we begin to explore the idea of multimodal imaging, let’s first start to explore the variety of preclinical imaging modalities that are most commonly used by researchers around the world.
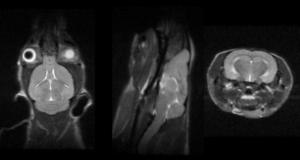
Modality: MRI
Available Now
MRI is considered the gold standard in soft tissue imaging, both in the clinic on patients and by researchers on a wide variety of preclinical imaging subjects.
Modality: CT
Available Now
Computed Tomography (CT) is one of the most commonly used clinical imaging techniques, next to perhaps ultrasound.
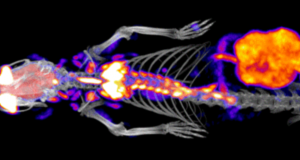
Modality: PET
Available Now
Nuclear imaging modalities include positron emission tomography (PET) as well as single photon emission computed tomography (SPECT), which both detect gamma radiation emitted from a radionucleotide injected into an imaging subject.
Modality: Ultrasound
Available Now
Ultrasound (US) imaging is one of the most commonly used diagnostic techniques clinically, and is widely used in preclinical imaging as well. It is a safe, non-invasive, and relatively inexpensive technology compared to some of the other available imaging modalities.
Modality: Optical
Available Now
When discussing Optical Imaging techniques in this series, the meaning is to cover bioluminescence (BLI) and fluorescence (FLI) imaging. T
Modality: Photoacoustic
Available Soon
Photoacoustic (PAI) imaging is a technology which combines the sensitivity of optical imaging with the depth of penetration and resolution of ultrasound imaging.
Modality: Microscopy
Available Soon