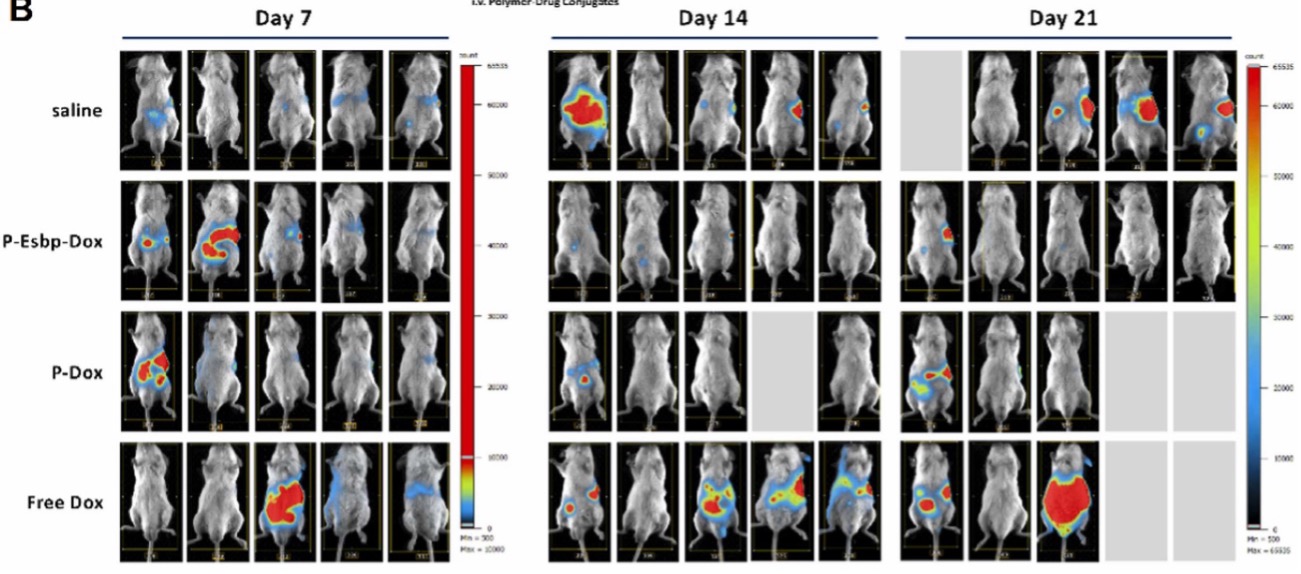
Cardiac disease is the main cause of death worldwide. Insufficient regeneration of the adult mammalian heart owing to the lack of cardiomyocyte proliferation and scarce therapeutic options for patients with advanced cardiac disease are major drivers of cardiac morbidity and mortality. Cardiac regeneration through cardiomyocyte proliferation occurs only in early postnatal mice but is lost shortly after birth when cardiomyocytes withdraw from the cell cycle 1,2.
Dr. Malek Mohammadi has recently established a neonatal model of transverse aortic constriction, to induce pressure overload in neonatal mice in the regenerative phase (postnatal day 1(P1) or in non-regenrative phase (P7). Her lab has demonstrated that neonatal mice at postnatal day 1 (P1) have the ability to adapt to pressure overload, but this ability is lost by P73. This adaptive mechanism in the regeneration phase (P1) is associated with preserved cardiac function, augmented cardiomyocyte proliferation and increased angiogenesis. Only seven days of pressure overload in the non-regenerative phase (P7), however, is associated with fibrosis in the heart and reduced cardiac function3,4. Thus, this fundamentally different response of the postnatal mouse heart to a pathological stimulus provides a model to explore the endogenous mechanism(s) underlying the adaptive and maladaptive response of the mammalian heart.
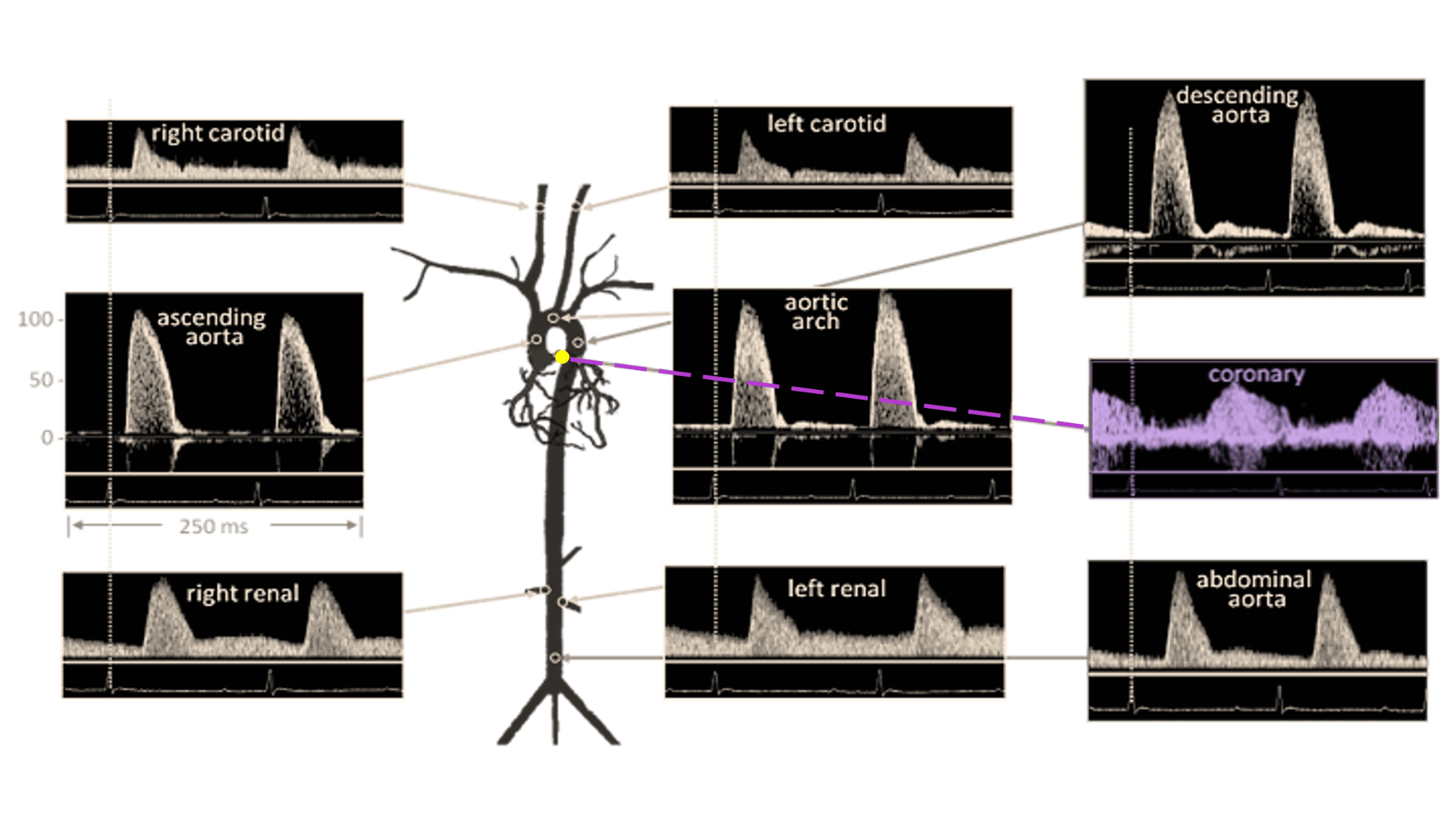
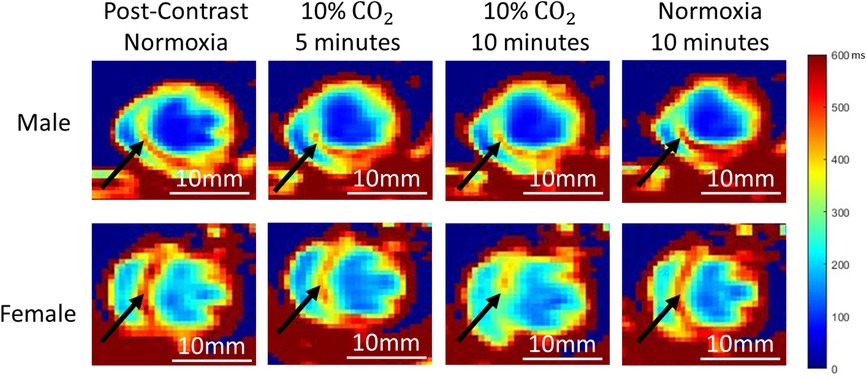
In this webinar, Dr. Malek Mohammadi provided an overview of the surgical protocol of neonatal model of transverse aortic constriction (nTAC) including postsurgical monitoring and ways to confirm the surgery using Doppler flow velocity measurements and echocardiography.
Furthermore, she discussed troubleshooting of some common problems that you might face during the surgery, and employing flow velocity and echocardiography.
The full protocol including video of the surgery is published at Nature Protocols and is available online: Malek Mohammadi, M., Abouissa, A.