System Used:
Pressure Arteriography

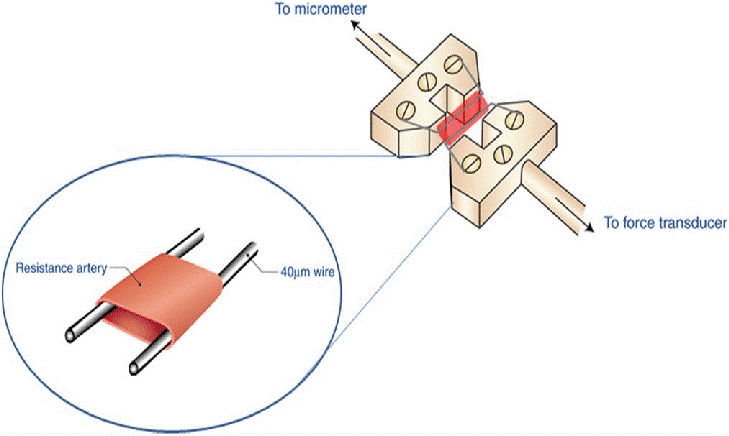
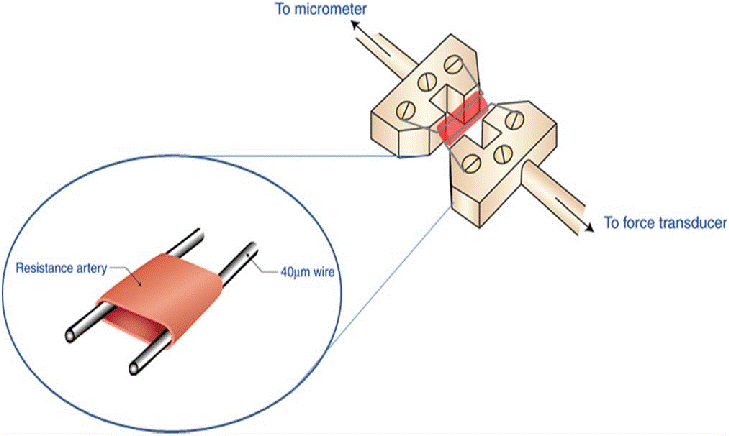
For those unfamiliar with vascular reactivity studies using a myograph, let’s discuss the basics of the protocol. A segment of the vessel of interest is dissected from an animal and placed into a dish containing a buffer solution to maintain tissue viability. Often, Krebs-Henseleit solution is used. Very carefully, using tissue forceps and scissors, tissue that has adhered to the vessel is removed. This must be done with great care to avoid damaging the vessel and impairing reactivity. Next, small sections of vessel are cut, and the wire or metal prongs are fed through the vessel and the apparatus is submerged in a bath chamber containing oxygenated buffer. The wire or metal prongs are then connected to a force transducer. When the vessel contracts or dilates, the force exerted on the wire or prongs provides a measure of reactivity.
The myograph is often used to assess endothelial dysfunction. One example is a study by Haj-Yehida and colleagues 1. The authors investigated the effects of a stable superoxide dismutase (SOD) mimic compound on endothelial dysfunction in diabetic rats. Following diabetes induction and administration of the SOD mimic compound for eight weeks, a small section of the thoracic aorta was dissected from the animal 1. Following preparation in Krebs-Henseleit solution, a 3 mm section was mounted to stainless steel wires and submerged in the bath chamber containing buffer 1. For wire myograph experiments, it is essential to use an oxygenated buffer, that contains electrolytes and glucose, heated to a physiological temperature to ensure tissue survival. The tissue must also be equilibrated at a resting tension with regular buffer changes prior to initiating a vascular reactivity protocol. In this study, phenylephrine was used to stimulate vasoconstriction and acetylcholine and nitroglycerin were used to stimulate vasorelaxation when added to the tissue bath 1. The response was used to generate a concentration-response curve for the sample, which revealed reduced effects of vasodilators in the diabetic compared to control animals.
Many wire myograph systems can be configured with wire or metal prong vessel mounts that enable the study of many different vessels. Depending on the configuration of the mount, small vessels from rodents as well as smaller vessels from large animals like pigs can be studied using a myograph.
A study by Rodríguez-Sinovas et al. examined whether ischemia-reperfusion injury alters coronary smooth muscle reactivity to vasoconstrictor stimuli such as methacholine using a pig model 2. Animals underwent a left anterior descending coronary artery occlusion for two hours, followed by a reperfusion period. The artery was then dissected and sectioned into thirds for assessment of regional differences in reactivity. Artery sections were mounted, submerged in the tissue bath containing Krebs-Henseleit buffer and equilibrated. Following contractions using 40 mM KCl, constriction response curves were generated using a methacholine stimulus (10-8 to 10-5 M, thirty minutes apart).
Peripheral vascular research is also a suitable application for the myograph. Puzserova et al. examined the effects of vascular aging on endothelial function if femoral arteries of normal and hypertensive rats 3. Femoral arteries were dissected from spontaneously hypertensive rats and Wistar Kyoto rats and mounted on a wire myograph system. Tissues were submerged in a bath containing a modified physiological salt solution oxygenated with 95% O2 and 5% CO2. Following normalization, arteries were precontracted with serotonin. Once steady-state contraction was reached, acetylcholine was introduced as a vasodilator to generate endothelium-dependent concentration-response curves. The authors also sought to determine the role of nitric oxide and administered NG-nitro-L-arginine methyl ester (L-NAME) to the bath prior to repeating the acetylcholine concentration-response curve.
Given these few examples, it is clear the wire myograph system enables countless physiological and pharmacological experiments. This versatile instrument is suitable for use with vessels ranging in size from small peripheral arteries to large vessels such as the aorta. There are some features to consider when exploring myograph instruments for your research:
- A removable mounting apparatus can enable the researcher to work with a microscope to mount especially small vessels. When threading a vessel onto the wires or prongs, it is essential to do so carefully to avoid damaging the lumen. Working under a microscope enables much more careful and accurate vessel preparations.
- Maintaining a stable buffer temperature for the vessel preparation is essential for accurate experiments. A circulating water bath provides an accurate means to control the temperature of your experiment.
- For pharmacology researchers working with costly drugs or compounds, the tissue bath size is a very important consideration. The ability to submerge the mounted vessel into a smaller bath reduces the amount of drug or compound necessary to achieve the desired concentration.
To learn more about all the myograph packages that Scintica offers click here
The wire myograph is a valuable tool for vascular researchers. I encourage you to explore the literature to learn more about the numerous applications for this instrument and contact us if you would like to discuss how to incorporate this technique into your research.
References:
- Haj-Yehia, A. et al. Effects of the SOD mimic nitroxide 3-carbamoyl-PROXYL on oxidative stress markers and endothelial dysfunction in streptozotocin-induced diabetic rats. Exp. Clin. Cardiol. 7, 85–92 (2002).
- Rodriguez-Sinovas, A. et al. Coronary smooth muscle reactivity to muscarinic stimulation after ischemia-reperfusion in porcine myocardial infarction. J Appl Physiol 95, 81–88 (2003).
- Puzserova, A., Ilovska, V., Balis, P., Slezak, P. & Bernatova, I. Age-related alterations in endothelial function of femoral artery in young SHR and WKY rats. Biomed Res. Int. 2014, (2014).