RFLSI-ZW Laser Speckle Contrast Imager
Robust Blood Perfusion Imaging System

System Overview
RFLSI-ZW laser speckle imaging system is an even better tool for microcirculation research based on laser speckle contrast imaging technology (LSCI).
With the advanced optical design and improved image processing algorithm, RFLSI-ZW shows greater performance in imaging field size, image quality, full-field frame rate and optical resolution, and provides a powerful and efficient means for human and animal tissue microcirculation measurement.
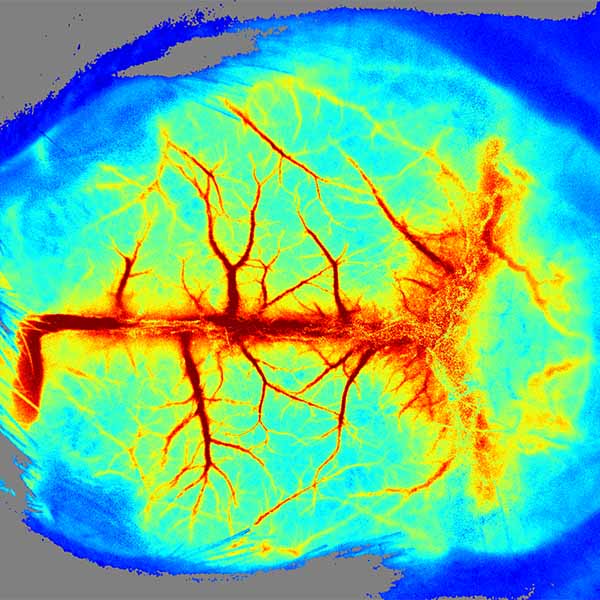
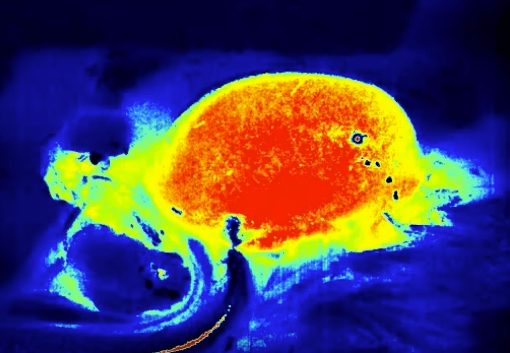
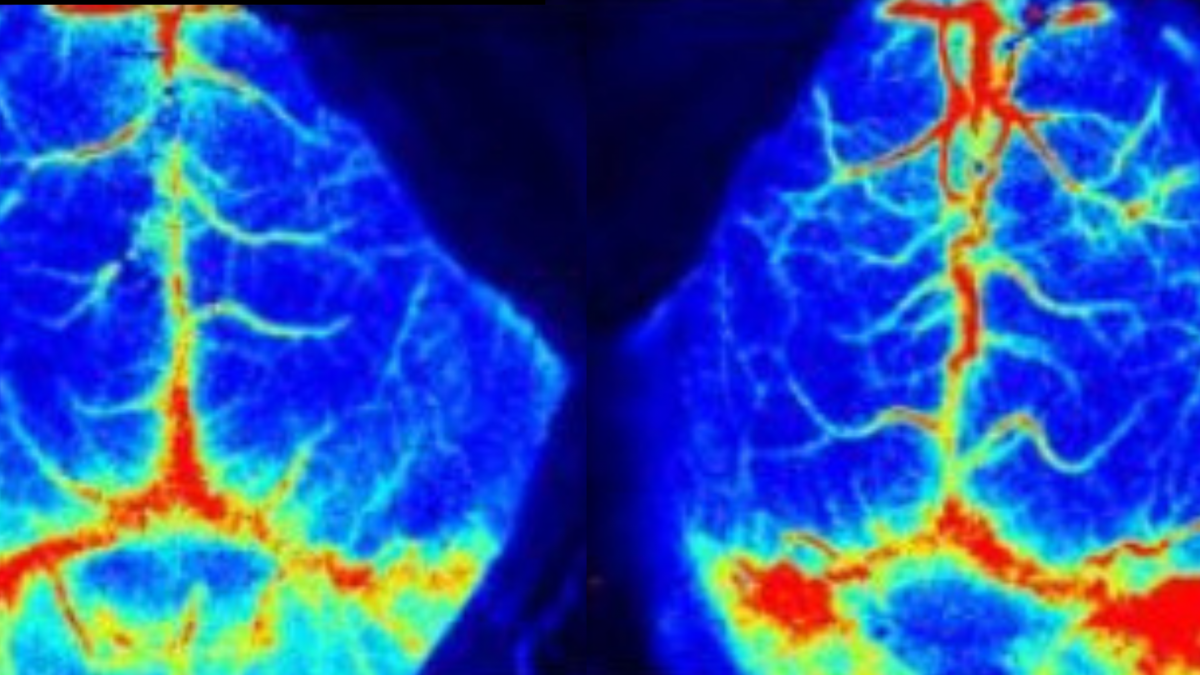
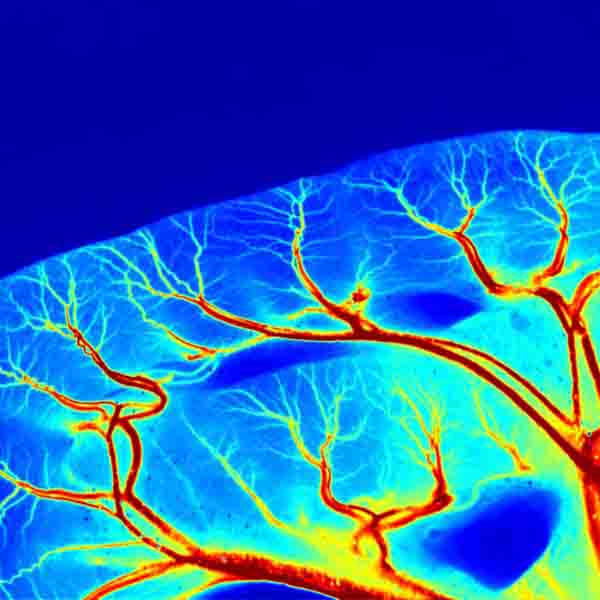
The LSCI technology advantages are its non-contact, high frame-rate, high spatial resolution. They can be used to observe and record blood perfusion of any exposed tissues or organs for microcirculation study or pre-clinical researches like ischemic stroke, lower limbs, mesentery, etc. Multi-Output includes blood perfusion images and videos (500+ million pixels), quantified data for perfusion unit and vessel diameter.
Performance Advantages
Image any exposed tissue (skin or surgically exposed tissues) and species.
Non-contact, non-contrast agent depending measurement.
- The built-in CMOS global shutter camera can achieve faster data acquisition and processing speed.
Best optical resolution of 3.9 μm/pixel, providing more detailed tissue structures.
Max frame rate (full field) up to 100 fps, acquiring real-time changes in larger areas.
Motorized 10x optical zoom and auto focus. Image size ranges from 0.57×0.75 to 22.5×30 cm2 in all-in-one imager, covering multiple research applications.
Fast auto and fine manual focus, improving focus efficiency and accuracy on various tissues.
Optimal lens assembly,filtering the ambient and reflecting light.
- Class 1 of measurement and indicating lasers, safe to use without eye protection System
Laser stability hardware for the ultimate in reliable and consistent measurement over minutes, hours and days.
Calibration with calibration box. Self-calibration is possible at any time to keep the equipment in optimal working condition.
Trigger In/Out BNC connections for communication with external devices.
Unlimited installation of analysis software in PC.
Features & Benefits
Reliable Data Measurements
Stable laser output, for reliable and consistent measurement over minutes, hours, and days, with accuracy up to 0.1 PU, makes it easy to control variables.
Quantified Visualized Data
Full-field monitoring with quantitative analysis. Multi-output includes vascular perfusion volume, vessel diameter, vessel angle, etc., also offline state analysis available.
Full-Frame HD Images & Video
High-definition camera (full-frame 2048 x 2048 resolution) helps you see the blood vessel endings and opens the world of microcirculation imaging.
Fast Data Acquisition
Data acquisition and blood perfusion imaging with video generated fast. USB 3.0 compatible.
High Frame Rate
A high-speed camera (up to 120 FPS) allows you to record rapid changes every second and record more details of blood vessel changes after treatment.
Easy Operation
Unique, no-contact technology design and no need for a special dark environment. The autofocus function is easy to use and gets perfusion data and video in seconds.
Technical Parameters
- Resolution
- Max Camera Resolution:2064×1544 pixels
Best Resolution:3.9 μm/pixel
- Max Camera Resolution:2064×1544 pixels
- Image:
- Flux/Gray/Intensity/Color/Overlay
Measurement Laser
785 m, Class 1
- Indicating Laser:
- 650 nmx2, Class 1
- Focus:
- Auto/Manual (fine focus)
- Trigger:
- 2×BNC
- Image Size
0.57× 0.75-22.5×30 cm2
- Max Frame Rate
100 FPS (full field)
- Zoom:
- 10x
- Working Distance
10-40 cm, continuous
- System Calibration
Calibration Box
- PC Connections
1 x USB 3.0 port
Software
Acquisition Software and Analysis Software
POWER SUPPLY
- Universal Voltage, 100V-230V. Note acquisition rate is unaffected by frequency of local electrical supply.
- Universal Voltage, 100V-230V. Note acquisition rate is unaffected by frequency of local electrical supply.
- Stand Options:
Scan head has standard VESA mount for desktop stand, Microstand and Clinical Mobile stand.
Applications
Cerebral blood perfusion monitoring
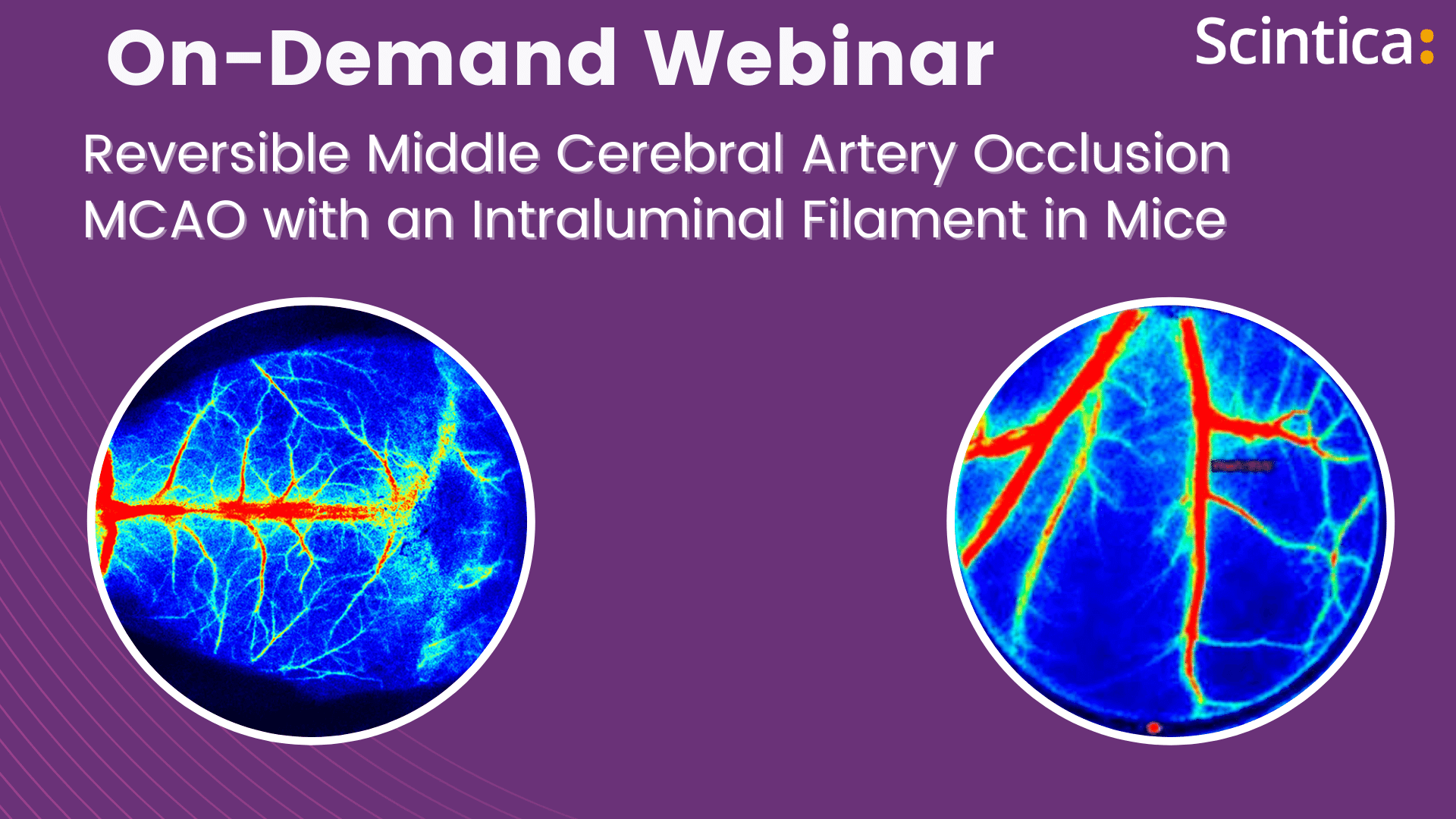
MCAO model assessment
Cortical spreading depression observation
Hind-limb ischemia research
Skin burn/skin flap transplantation
Organ microcirculation observation
Skin allergies
Septic Shock
Chicken Chorioallantoic Membrane Assay
Diabetic Foot
Laser Speckle Imaging Gallery
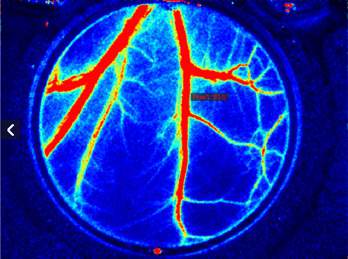
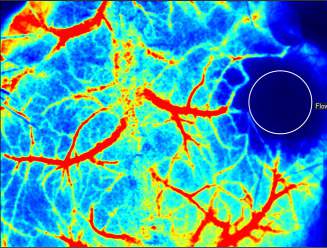

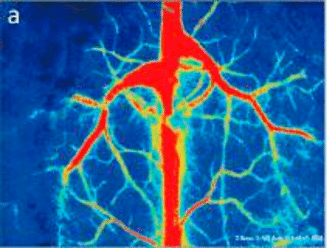
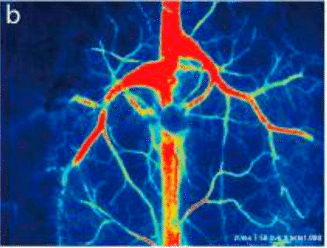
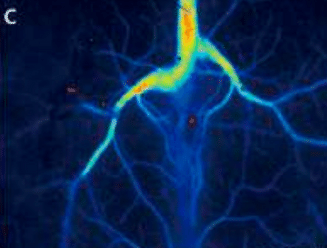
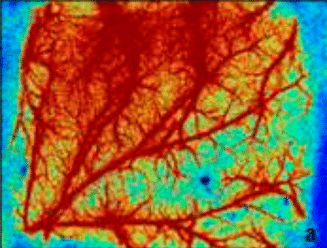
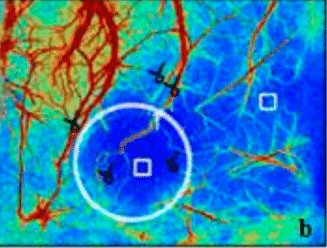
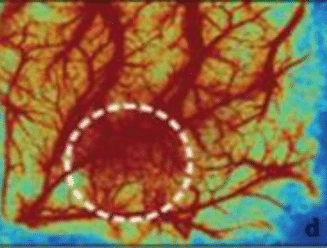
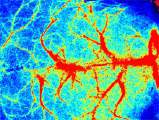

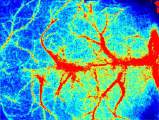

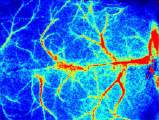
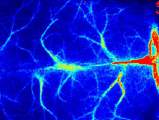
Past Webinars - RFLSI III Laser Speckle Perfusion Imager
Publications & Articles
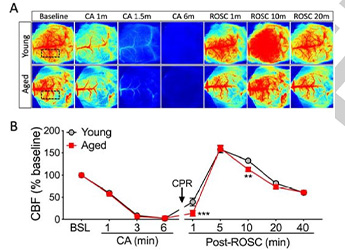
Detect the Changes of the Cerebral Cortex Blood Perfusion of a Cardiac Arrest (CA) and Return of Spontaneous Circulation (ROSC) Mouse Model
Detect the Changes of the Cerebral Cortex Blood Perfusion of a Cardiac Arrest
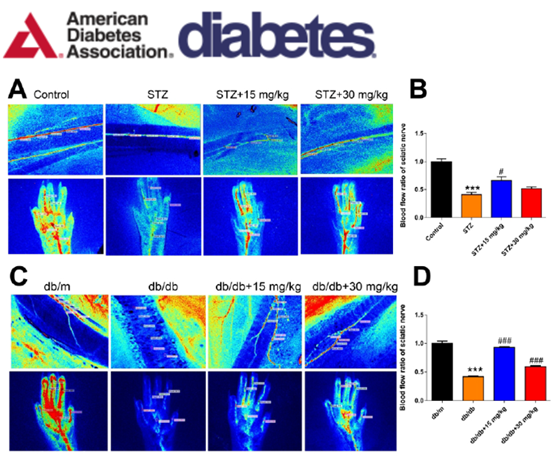
Real-Time Monitoring of the Regional Velocity, Distribution of Blood Flow, and Perfused Vessel for Sciatic Nerve and Foot Pads
Real-Time Monitoring of the Regional Velocity, Distribution of Blood Flow, Perfused Vessel for
Using the Laser Speckle to Observe Changes in Cerebral Blood Flow after Lymphatic Ablation and Quantitative Analysis
Using the Laser Speckle Imaging System to Observe Changes in Cerebral Blood Flow