TriTom™
Small Animal Whole Body Photoacoustic and Fluorescence Molecular Imaging Platform

System Overview
The TriTom imaging platform utilizes photoacoustic and fluorescence tomographies (PAFT) to enable high-resolution (up to 160 µm) non-invasive in vivo whole-body imaging in small animal models. With the ability to use multiple excitation wavelengths per scan, the multi-modality system can simultaneously acquire photoacoustic and fluorescence data in large volumes (> 25 cm3) allowing for spectroscopic molecular analysis within the region of interest. In addition to 3D molecular maps, the TriTom enables spatially-resolved assessment of physiologic parameters in vivo, such as volumetric blood content and oxygenation without the need for contrast agents. The TriTom also provides quantitative imaging for a wide range of fluorophores and other types of molecular probes excited between 460 nm and 1320 nm. With such versatile imaging capabilities, the state-of-the-art TriTom system is ideally suited for a wide range of preclinical applications including cancer, toxicology, developmental biology, tissue engineering and regeneration, neuroscience, cardiovascular imaging, as well as in the development of drugs, therapies, and optical and fluorescent imaging probes.
The TriTom is specially designed for imaging small animal models with a convenient animal handling set-up for fast, reproducible positioning of the subject and integrated gas anesthesia to quickly move from animal preparation to imaging. The rapid image acquisition time (~36 seconds) allows for fast imaging of individual subjects or capturing dynamic processes such as biodistribution, lymphatic drainage, or vasodilation. Additionally, the temperature control unit maintains the imaging environment within ± 0.1°C, ensuring the animal’s welfare throughout the imaging session.
A user-friendly image acquisition software enables new users to start collecting data quickly while maintaining the flexibility for advanced users to optimize imaging parameters for their specific research applications. The image reconstruction software is also designed for rapid reconstruction and visualization of large volumes with an open data format that allows for flexibility in image reconstruction and data management. With such versatile imaging capabilities, the state-of-the-art TriTom system is ideally suited for a wide range of preclinical applications including cancer, toxicology, developmental biology, tissue engineering and regeneration, neuroscience, cardiovascular imaging, as well as in the development of drugs, therapies, and optical and fluorescent imaging probes.
Features & Benefits
Combined 3D photoacoustic and fluorescence tomography
By combining both 3D photoacoustic and fluorescence tomographies the TriTom system provides:
- Deep tissue quantitative imaging (no depth limits in mouse models) with a high spatial resolution (up to 160 µm).
- Superior molecular sensitivity.
- Accurate 3D registration of anatomical, functional, and molecular volumes.
- Accurate registration (in space and time) of multimodality (photoacoustic and fluorescence) images.
Single-scan assessment of multiple microsamples of contrast agents
Accelerates the development of novel contrast agents and molecular probes without wasting expensive samples.
Single-scan assessment of multiple (up to 10) microsamples (50 µL or less) of contrast agent quick-loaded into microcuvettes.
Integrated software suite
- TriTom’s user-friendly interface minimizes the learning curve for operating the system and allows for easy image acquisition using quick-start presets or customizable scanning procedures.
- Large-scale volumes and molecular maps are reconstructed in seconds with established procedures for image processing and analysis in open-source software (3D Slicer).
- The open data format also allows for image reconstruction and data management with third-party software.
Fast imaging scans of large volumes
The short scan time on the TriTom enables assessment of slow dynamic processes (time constants ≥1 min) such as biodistribution of molecular probes, lymphatic drainage, vasodilation, etc.
- Fast (<36s) imaging scans of large (> 25 cm3) volumes.
Nanosecond pulsed laser
Enables quantitative photoacoustic and fluorescence imaging with a very broad assortment of endogenous molecules (hemoglobin, water, melanin, lipids, etc.), molecular probes (fluorophores, nanoparticles, quantum dots) with excitation from visible to NIR-II range.
- Tunable in a wide spectral range: 460 nm – 1320 nm.
Multi-modality compact design
Saves valuable laboratory space.
High-sensitivity sCMOS fluorescence camera combined with a nanosecond pulsed laser excitation
High signal-to-noise ratio of fluorescent images acquired with short exposures (10-20 ms) enables high frame rate image acquisition (up to 35 fps), fast 3D scans, and can be used to study dynamic rapid processes.
Optical excitation wavelength is tunable
Enables fast molecular imaging scans, which require multiple excitation wavelengths (unmixing multiple molecular species using both photoacoustic and fluorescence imaging channels).
Optical excitation wavelength is tunable across the entire spectral range (460 nm – 650 nm and 650 nm – 1320 nm windows) with speeds up to 20 Hz.
Integrated gas anesthesia line and adjustable mouse holder
Designed for convenient operation and repeatable in vivo longitudinal studies.
Up to 360° rotational coverage of the subject during a scan
True 3D reconstruction of the sample and high-resolution axial, coronal, and sagittal images on the TriTom system.
Temperature control unit
Maintains the temperature of the sample environment within ± 0.1°C.
Models & Specifications

TRITOM - Standard
-
Photoacoustic (PA) Imaging Channel
-
Fluorescence (FL) Imaging Channel
-
4 Standard Emission Filters
-
sCMOS Camera
-
Control Station
-
Image Acquisition Unit
-
Laser Excitation Unit (532 nm & 640-1300 nm)
-
Excitation Fiberoptic Bundle
-
Accessories

TRITOM - Premium
-
Photoacoustic (PA) Imaging Channel
-
Fluorescence (FL) Imaging Channel
-
8 Standard Emission Filters
-
High Sensitivity sCMOS Camera
-
Control Station
-
Image Acquisition Unit
-
Laser Excitation Unit (460 -1300 nm)
-
Excitation Fiberoptic Bundle
-
Accessories
Accessories & add-ons
Microcuvette Sample Holder
Designed to aid in the development of molecular probes, optical contrast agents, and fluorophores. This accessory allows the user to interrogate up to 10 samples at a time and requires ≤ 50 μL sample volumes. The samples are easily loaded into the radial slots of the Sample Holder and the holder is mounted in the TriTom imaging system using magnetic connections allowing for convenient and repeatable data collection.
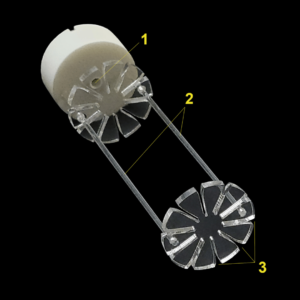
(1) Port for administering liquid scattering background,
(2) plastic support rods
(3) radial slots for quick setup and removal of microcuvettes
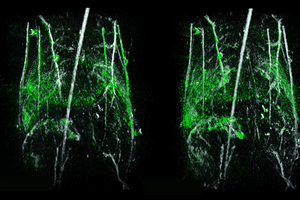
Mouse Holder
Designed to provide accurate and repeatable mouse positioning, which is essential for longitudinal in vivo imaging. This accessory not only secures the animal in place using cushioned paw mounts and a bite bar but also allows for the critical delivery of anesthesia gas to the animal during the scan. The Mouse Holder is easily attached to the TriTom for imaging.
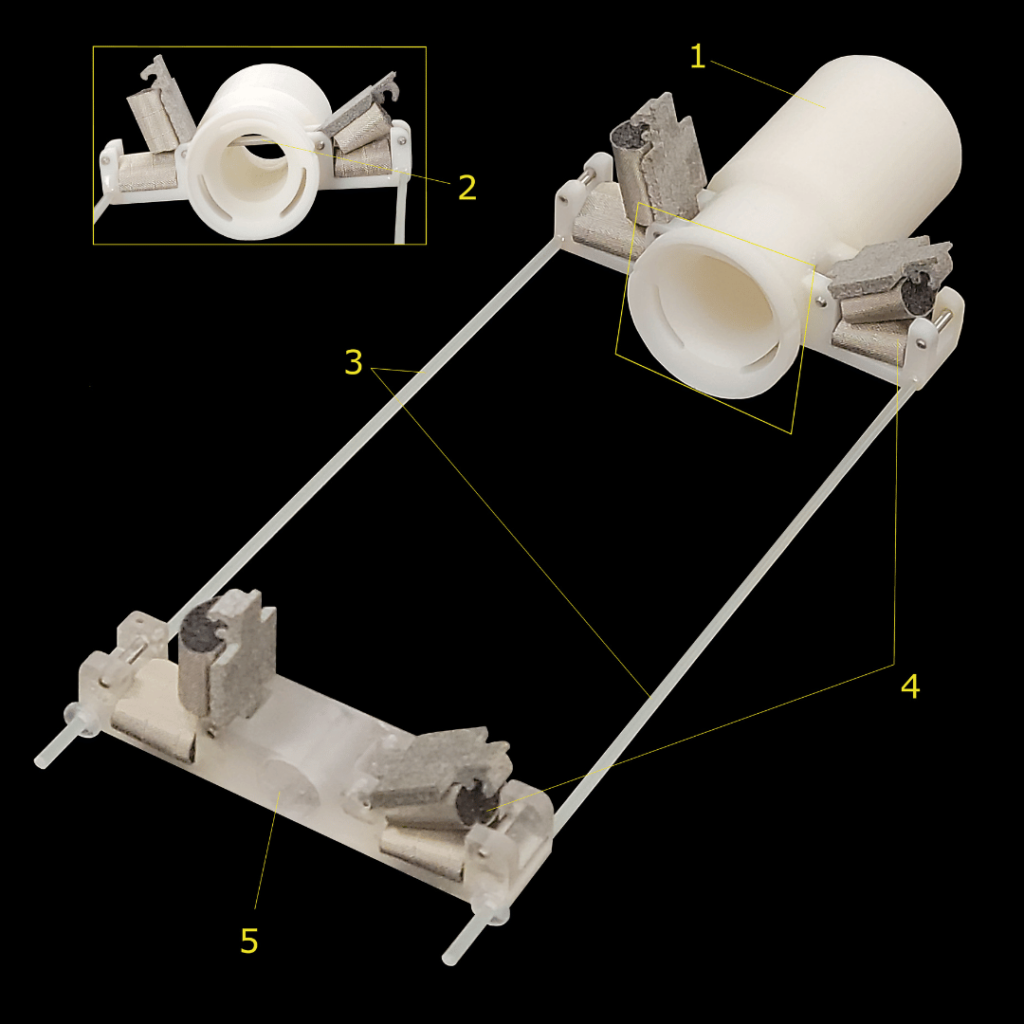
Mouse holder (1) Hollow shaft to fix the mouse holder inside the TriTom while reliably delivering anesthesia gas, (2) bite bar for secure and repeatable mouse positioning, (3) plastic support rods, (4) cushioned paw mounts, and (5) adjustable support block for mouse’s hind legs.
Applications
Anatomical Imaging / Registration
The TriTom provides high-fidelity, noninvasive whole-body anatomical imaging of small animals showing the details of the entire cardiovascular system and internal organs with spatial resolution up to 160 µm.
TriTom imaging takes advantage of the intrinsic optical properties of tissue, specifically hemoglobin, to provide high-resolution images of both superficial and deep blood-rich structures in murine models, which is crucial for understanding the fundamental relationship between constructed molecular maps and anatomical structure.
In addition to whole-body anatomical imaging, the TriTom can be utilized for functional imaging of physiological processes, which can include monitoring blood oxygenation, tumor growth, or evaluating therapeutic effects.
The high-resolution TriTom reconstructions also enable visualization of deep tissue structures with extraordinary detail, enabling superior anatomical reference.
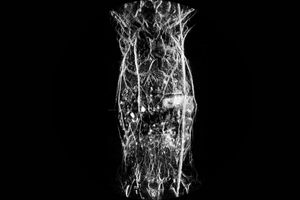
Whole-body in vivo Anatomical Imaging
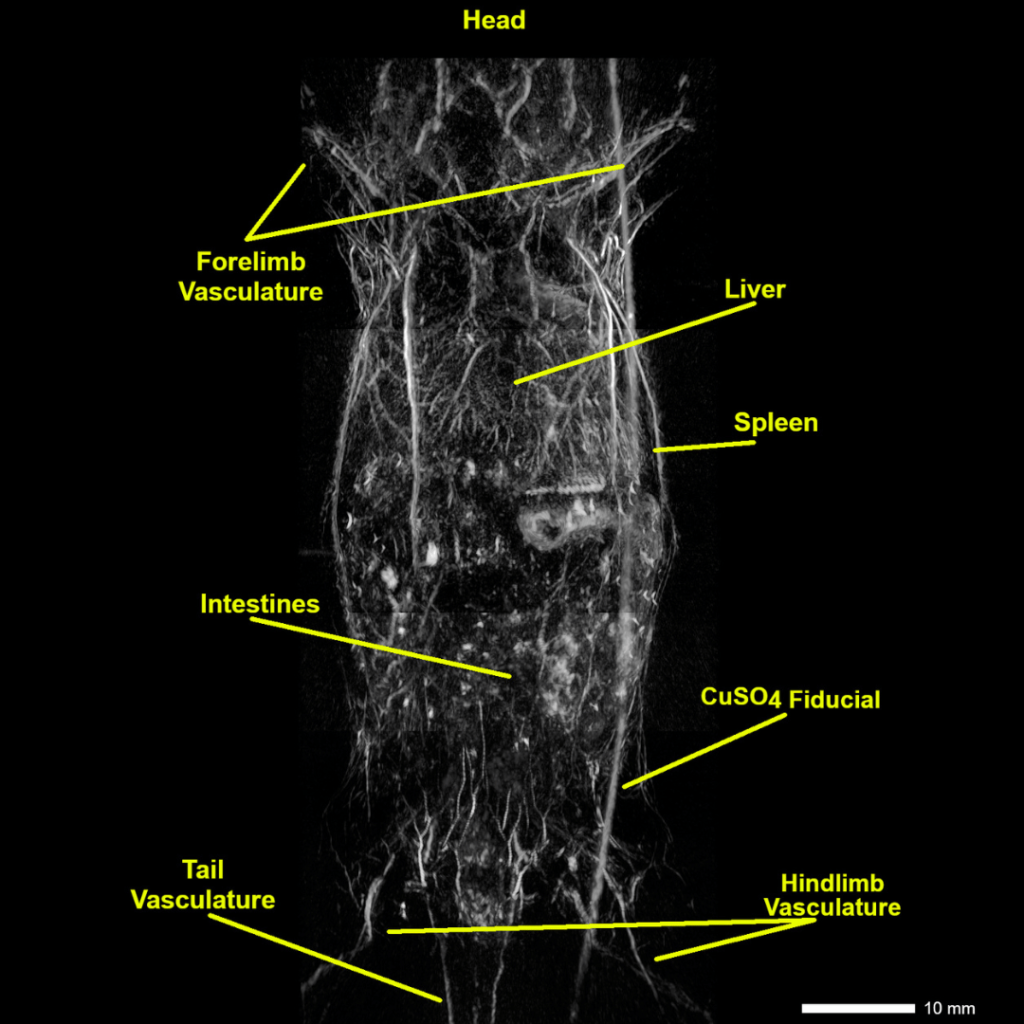
Whole-body reconstruction of a nu/nu nude mouse. Image acquired with NIR 1 excitation at 800 nm.
Imaging Deep Anatomical Structures
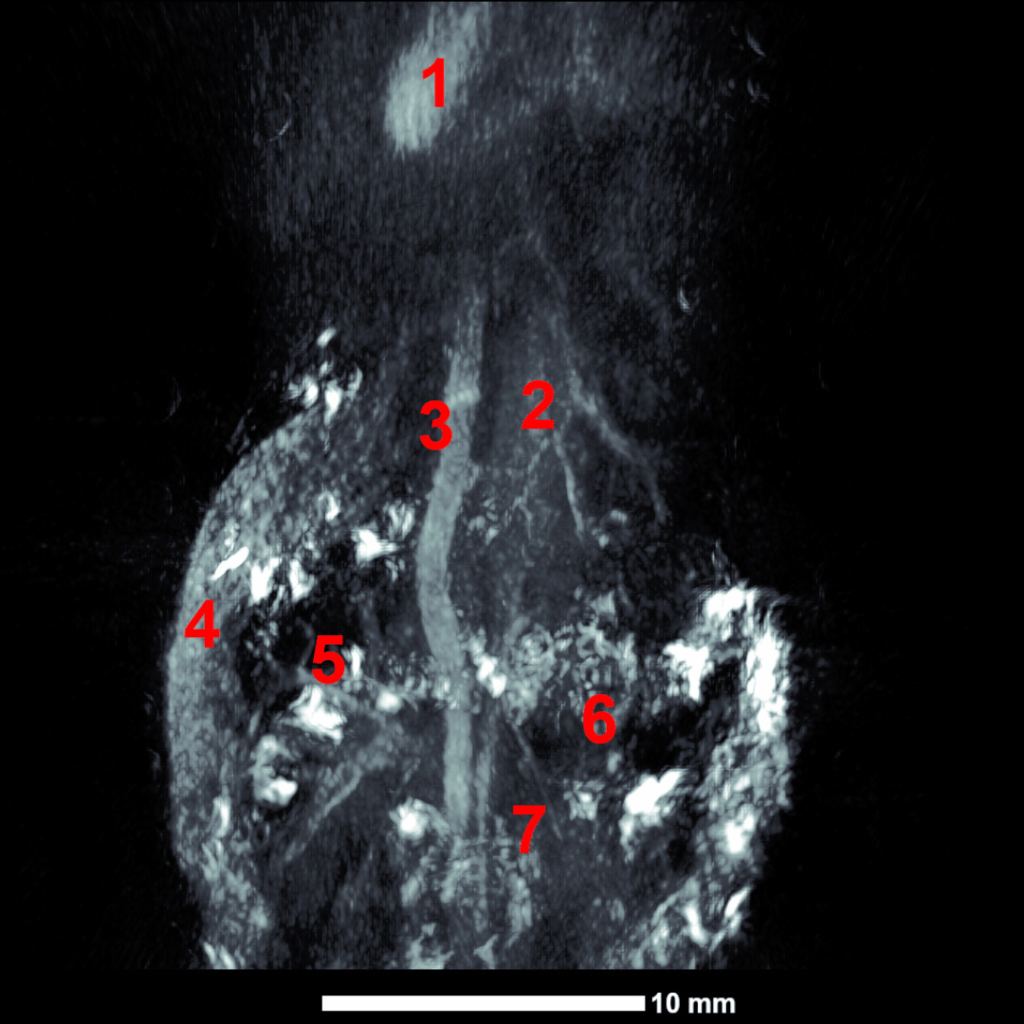
Mouse thorax and upper abdomen . (1) heart, (2) liver, (3) aorta, (4) spleen, (5) left kidney, (6) right kidney, and (7) intestines. Image acquired with high-energy NIR 1 excitation at 1064 nm.
Superior Anatomical Reference
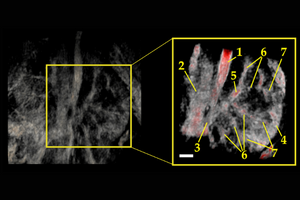
Mouse Kidney; (1) abdominal aorta, (2) inferior vena cava, (3) renal vein, (4) renal cortex, (5) renal artery, (6) interlobular artery (vein), and (7) medulla (renal pyramids). Scale bar = 1 mm.
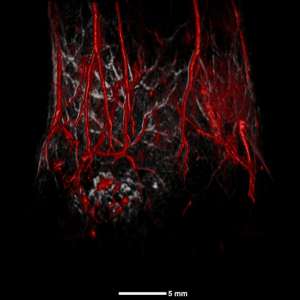
Cancer
The TriTom can be used for the in vivo assessment of tumor growth, detection and monitoring of metastatic disease, and characterizing the tumor microenvironment, such as hypoxia and blood perfusion. The high sensitivity of TriTom imaging can accurately detect contrast agents targeted to tumor biomarkers as well as the tumor response to cancer therapies.
The tracking of a contrast agent’s lymphatic drainage enables further case studies of metastasis progression in lymph nodes, which is crucial for evaluating cancer staging and prognosis in the development of anticancer drugs and therapies. Malignant primary tumors spread cancerous cells by metastasizing through the lymphatic system or through the vasculature.
In addition to whole-body anatomical imaging, the TriTom can be utilized for functional imaging of physiological processes, which can include monitoring blood oxygenation, tumor growth, or evaluating therapeutic effects.
The high-resolution TriTom reconstructions also enable visualization of deep tissue structures with extraordinary detail, enabling superior anatomical reference.
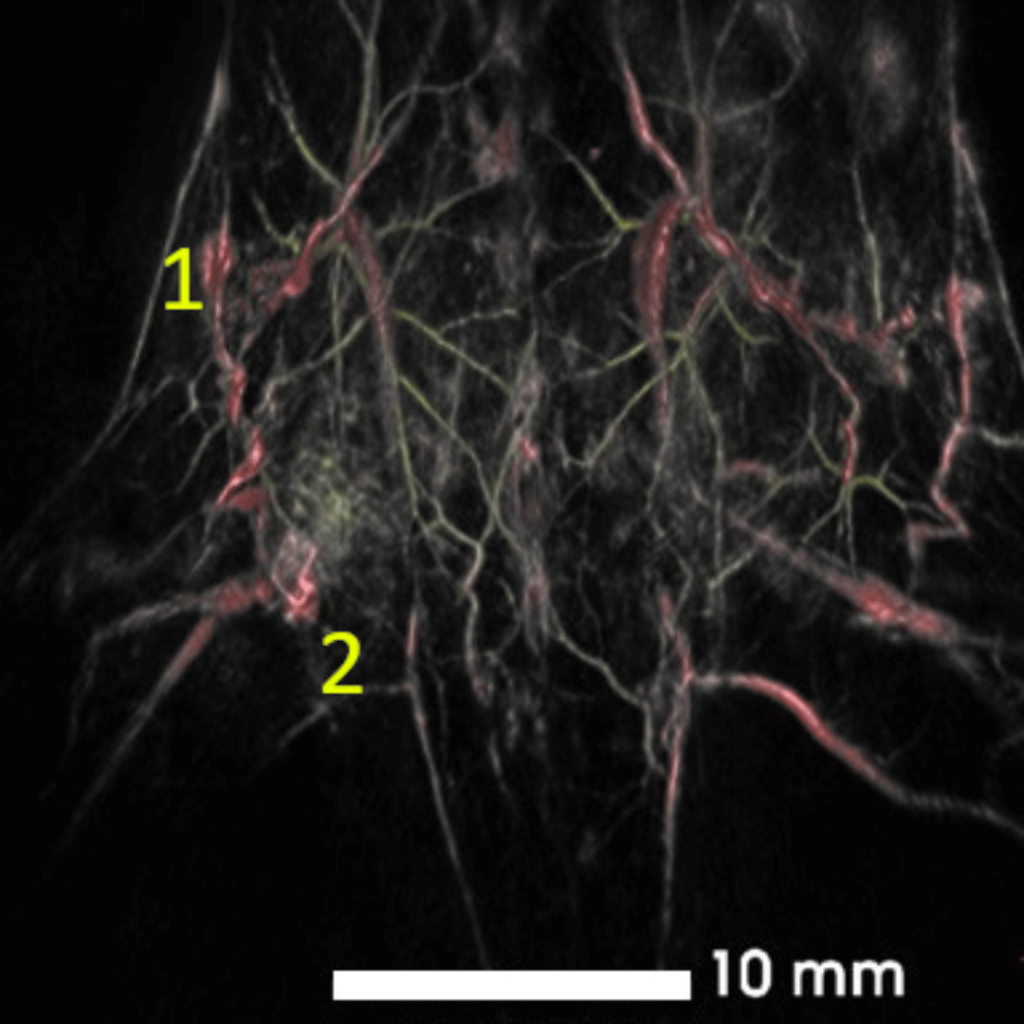
Human breast ductal carcinoma xenograft (BT474 cells) in nu/nu mice. Composite image acquired with visible (532 nm, yellow) and NIR I excitations (890 nm, red/grey) showing superficial and deep tumor vasculature.
Tumor size = 10.6 x 4.7 x 11.6 mm3
Reference:
Dumani et al, Proceedings SPIE 10878, Photons Plus Ultrasound: Imaging and Sensing, 108784Y (2019) »
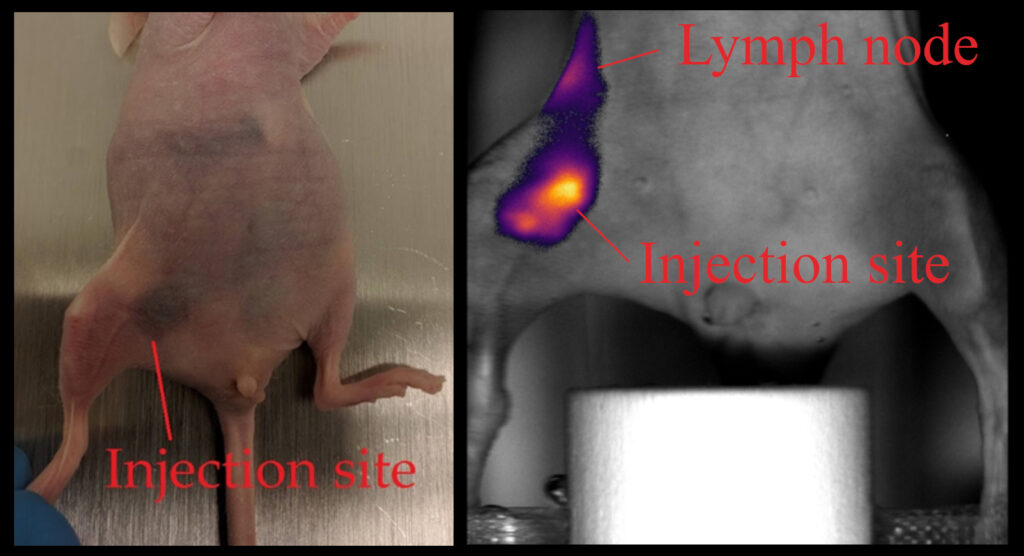
Normal mouse, 24-hrs post-subcutaneous injection of glycol-chitosan-coated gold nanospheres (50 µg/mL) mixed with ICG (50 µg/mL). The total injection volume is 50 µL. Images were acquired with NIR I excitation at 770 nm.
Thompson, W. R., Brecht, H.-P. F., Ivanov, V., Yu, A. M., Dumani, D. S., Lawrence, D. J., Emelianov, S. Y., & Ermilov, S. A. (2023). Characterizing a photoacoustic and fluorescence imaging platform for preclinical murine longitudinal studies. Journal of Biomedical Optics, 28(03). https://doi.org/10.1117/1.JBO.28.3.036001
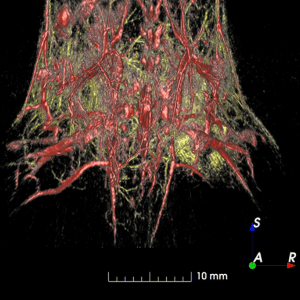
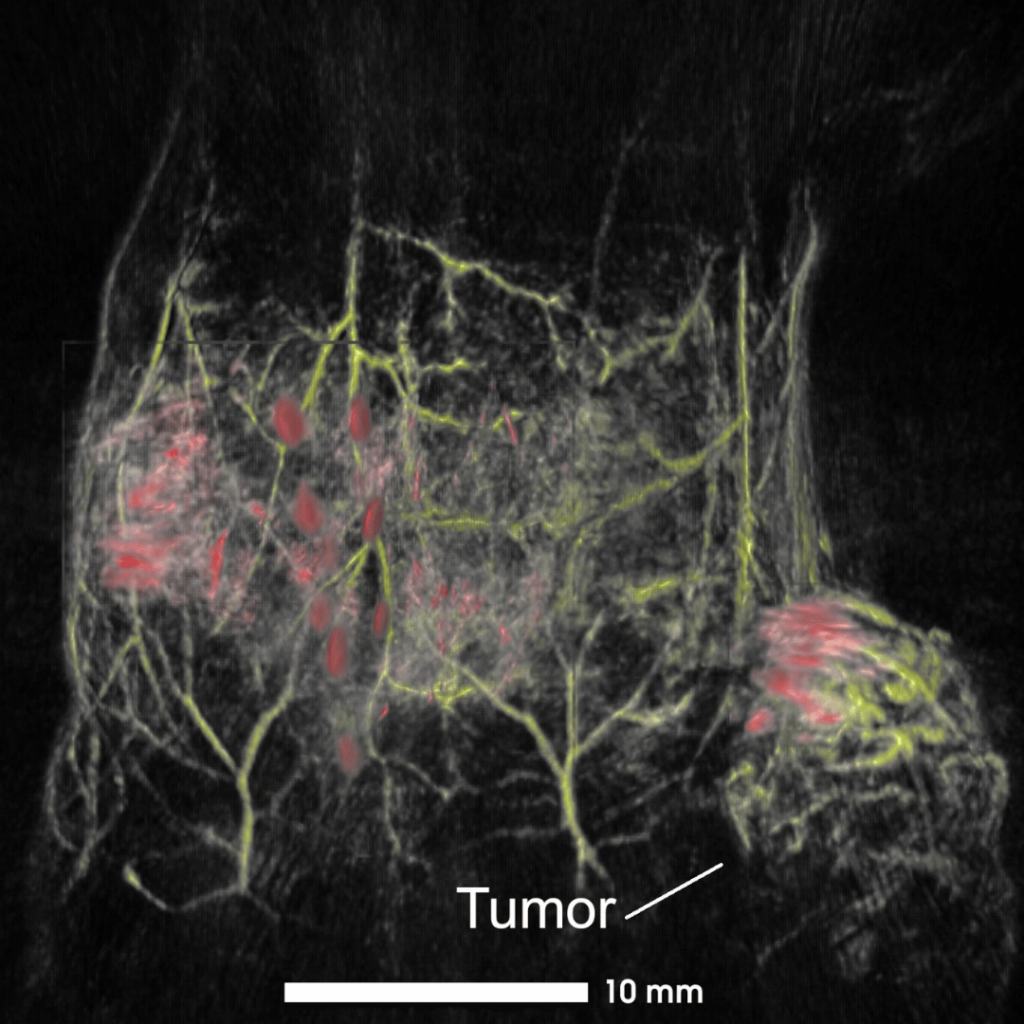
Lower abdomen of a healthy mouse showing deep vascular networks and contrasted lymphatics. Composite image acquired with visible (532 nm, yellow) and NIR I excitations (890 nm, red/grey). (1) right subiliac lymph node and (2) contrast injection site (right mammary fat pad).
Tissue Engineering and Regeneration
The high molecular sensitivity of the TriTom enables whole-body assessment and monitoring, which is a significant advantage for tissue engineering applications such as tracking stem cell migration in vivo . The TriTom produces accurate and quantitative 3D images with submillimeter spatial resolution that can be efficiently analyzed by virtual sectioning in any of the standard (axial, coronal, and sagittal) or non-standard anatomical planes. This unique characteristic of 3D TriTom images is beneficial for applications where the target anatomy and regions of interest are difficult to identify in a single anatomical view.
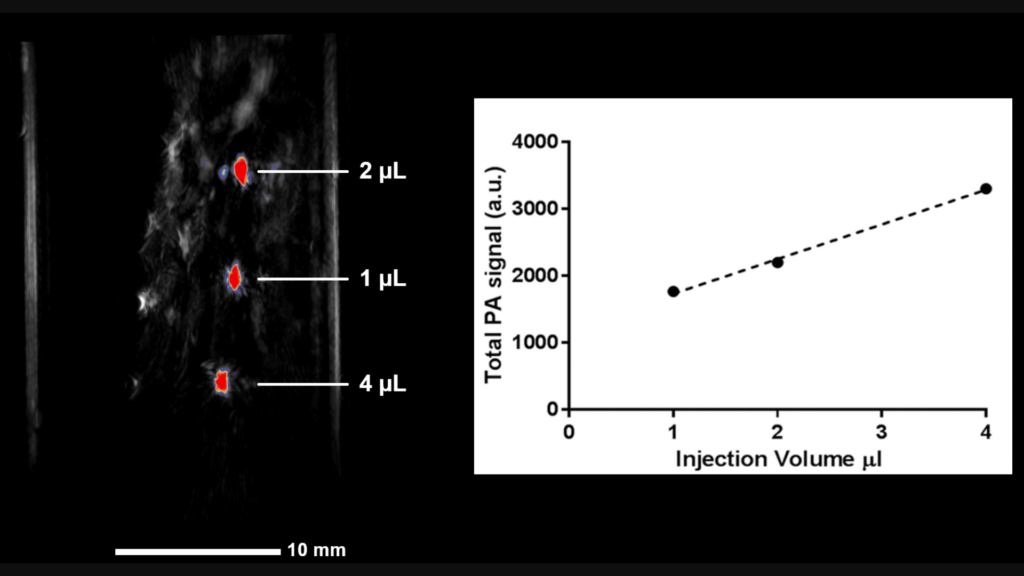
A TriTom image (3D rendering) of gold nanosphere-labeled mesenchymal stem cells locally injected in a rat spinal cord. The linear relationship between the average image intensity and injection volume demonstrates the ability of the TriTom to track and monitor cell migration in vivo. Scale bar = 10 mm.
Reference: Donnelly et al, Nano Letters, 18, 6625-6632 (2018)
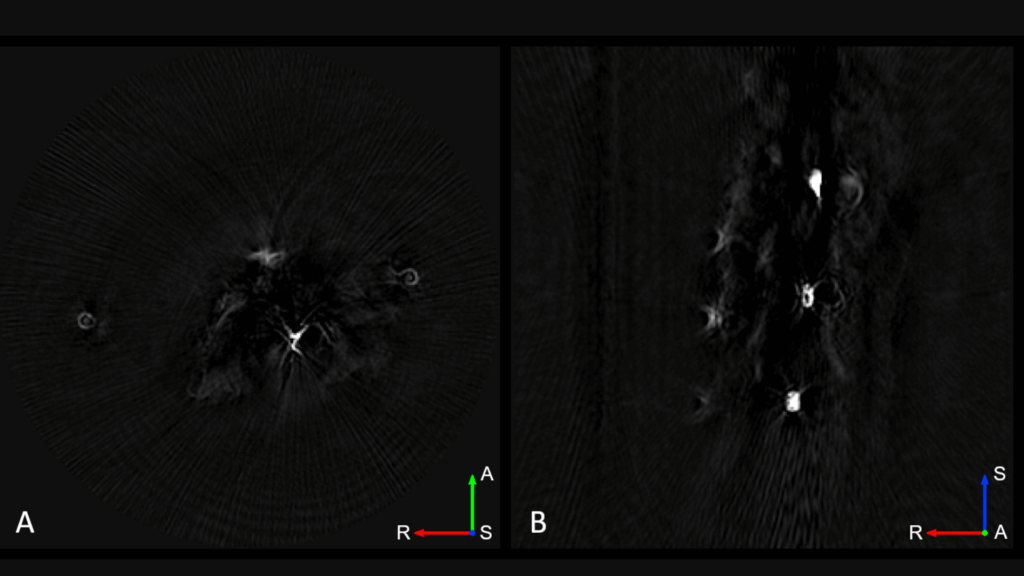
The TriTom’s submillimeter spatial resolution in all three anatomical planes makes image analysis and quantification accurate and easier for applications where small anatomical structures are challenging to identify.
Reference: Donnelly et al, Nano Letters, 18, 6625-6632 (2018)
Developmental Biology
The TriTom provides large-scale volumetric images which can be used to assess the stage and development of multiple embryos in a single image acquisition. The high spatial resolution enables studies of complex 3D vascular structures, such as the uterine and spiral arteries, which are challenging to assess with traditional 2D imaging methods.
Additionally, the TriTom small animal imaging platform can be used for functional imaging of pregnancy-related disease indicators, such as blood oxygen saturation and targeted imaging of developmental biomarkers. The exceptional spatiotemporal resolution of the system can also be used to detect the dynamic response to therapies.
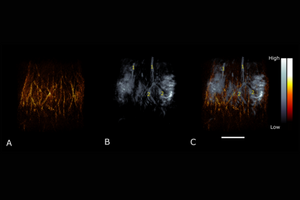
3D renderings of a pregnant mouse on gestational day 12 presented as (A) superficial, (B) deep tissue, and (C) a composite of superficial (orange) and deep tissue (gray) images. The (1) common iliac arteries and (2) placentas are labeled. Scale bar = 10 mm.
Reference: Huda et al., Photoacoustics, 100209 (2020)
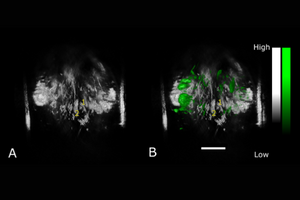
TriTom images of a pregnant mouse on gestational day 17 A) pre-injection, (B) 5 minutes post-injection, and (C) 90 minutes post-injection of folate-targeted ICG. The 1) placenta and (2) spiral artery are labeled. Scale bar = 10 mm.
Reference: Huda et al., Photoacoustics, 100209 (2020)
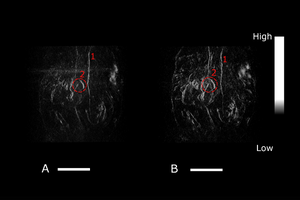
Deep-tissue TriTom image (NIR I excitation at 808 nm) of the abdomen of a pregnant mouse A) pre-injection and (B) 13 minutes post-injection of a vasodilator.. (1) Denotes the iliac artery, and (2) denotes the placenta. Scale bar = 10 mm.
Reference: Huda et al., Proc. SPIE 11960. March (2022)
Neuroimaging
The TriTom imaging system includes a mouse holder with a design optimized for non-invasive in vivo brain imaging. The submillimeter spatial resolution of the TriTom enables detailed visualization of vascular structures within the brain while the fast image acquisition time can capture dynamic physiological processes in vivo, such as penetration of the blood-brain-barrier (BBB) by drugs and their accumulation in local areas of the brain.
Brain Imaging – Application Note

A composite TriTom image of the brain and neck of a female BALB/c mouse acquired post-mortem with visible (532 nm, orange) and NIR I (750 nm, gray) excitation wavelengths.
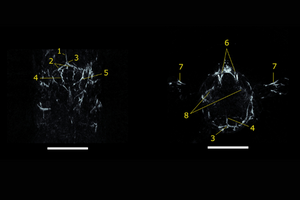
10 mm-thick (A) coronal and (B) transverse maximum intensity projection slabs of a mouse brain acquired with NIR I (750 nm) laser excitation. (1) Superior sagittal sinus, (2) transverse sinus, (3) confluence of sinus, (4) cerebral artery, (5) auricular artery, (6) jugular vein, (7) brachial artery, and (8) ophthalmic artery. Scale bar = 5 mm.
Development of molecular probes, optical contrast agents, and fluorophores
A critical step in the development of novel contrast agents is determining the optical absorption, fluorescence, and/or photoacoustic spectrum of the sample. The TriTom can expedite the progression of contrast agent development from characterization to in vivo applications via high throughput, quantitative imaging. Further, the microcuvettes provided with the system only require a small volume (≤50 µL) of each sample and up to 10 samples can be analyzed simultaneously; this is especially beneficial with difficult to produce or expensive contrast agents.
A key advantage of the TriTom imaging platform is the combination of deep-tissue imaging, high spatial resolution, and high molecular sensitivity, which enables 3D maps of molecular or functional processes accurately registered with animal anatomy. The TriTom is able to accurately differentiate exogenous contrast agents from background tissue and blood using a multispectral molecular unmixing algorithm for holistic analysis in vivo.

Transverse TriTom images. The samples in microcuvettes scanned with (A) 720 nm and (B) 810 nm excitation wavelengths are hard to differentiate in contrary to images obtained after the application of a multispectral unmixing algorithm (C) and (D). The volumetric ratios of NiSO4 to CuSO4 are as follows: (1) [1:0], (2) [3:1], (3) [1:1], (4) [1:3], and (5) [0:1].
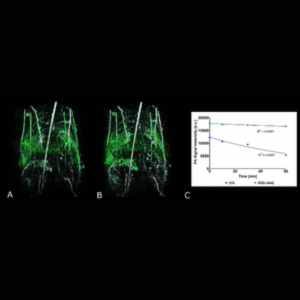
Molecular imaging of an ICG-based contrast agent (green) overlaid on the anatomical image of a mouse body (NIR I excitation at 800 nm, white) at 0, and 90-min post-injection. The graph shows the circulation half-life of the ICG contrast agent compared to ICG.
Reference: Singh et al., Photoacoustics, December (2022)