Article Review: A Surrogate Marker for Very Early-Stage Tau Pathology is Detectable by Molecular Magnetic Resonance Imaging
System Used:
M-Series Compact MRI

There is a critical need to reduce the burden of neurodegenerative diseases on healthcare systems. Alzheimer’s disease and Parkisons’ diseases are some of the conditions associated with escalating financial pressures and very limited treatment options. Improving the early diagnosis rate could be key to implementing measures to reduce and prevent further progression of the disease, as well as provide new insight into the underlying biology of neurodegeneration.
A widely established feature of many neurodegenerative diseases, including Alzheimer’s disease (AD), and Parkinson’s disease (PD) is the presence of intracellular aggregated filamentous tau protein (collectively termed “tau-opathies”). The abnormal phosphorylation of tau protein is a necessary precursor to the formation of tau fibrils and results from a pathological hyperphosphorylative neuronal state.
In this study, Parekh and colleagues identified translocated cell-surface bound vimentin as a unique cell surface marker of the hyper-phosphorylative neuronal state (vimentin is an intracellular protein in normal conditions) in a cell-based model of tau pathology. The author designed and validated an MRI-based molecular imaging strategy based on the use of PEGylated liposomes bearing macrocyclic Gd-chelates targeted at vimentin via DNA adaptamers (TauX-NP) or small molecule vimentin ligand warfarin A (WNP).
The authors subsequently used our Aspect Imaging M-Series compact M7 MRI scan and T1-weighted sequences to validate their approach in vivo in the P301S transgenic murine model of abnormal Tau at 2 months of age, when the transgenic mice do not exhibit frank Tau pathology (pre-pathological stage). Both the TauX-NP and WNP produced signal enhancement on MRI in the signal enhancement in the cortex and the hippocampus regions of the brain brains of transgenic mice, 4 days after intravenous injection of the nanoparticles but not in the brains of non-transgenic siblings. Untargeted nanoparticles did not show any signal enhancement in either group of mice. Practically 100% of the P301S mice go on to develop frank tau pathology at 8 months of age or later. This was further corroborated and validated in the APP/PSEN1 transgenic mouse models of AD.
In summary, Parekh and colleagues have designed and developed a molecular imaging strategy able to detect cell surface marker hyperphosphorylative neuronal state – a state which coincides with the initiation of a decades-long process culminating in AD. This study warrants the clinical evaluation of this MRI-based molecular approach as it could advance the diagnosis of neurodegenerative disease by several years.
The field strength of the M-Series (1 Tesla) is optimal for the development of clinically translatable functional and molecular imaging approaches and for ensuring higher signal enhancement with molecular probes using gadolinium as a source of MRI.
Please contact us at Scintica Instrumentation to learn more about the Aspect Imaging M-series MRI systems.
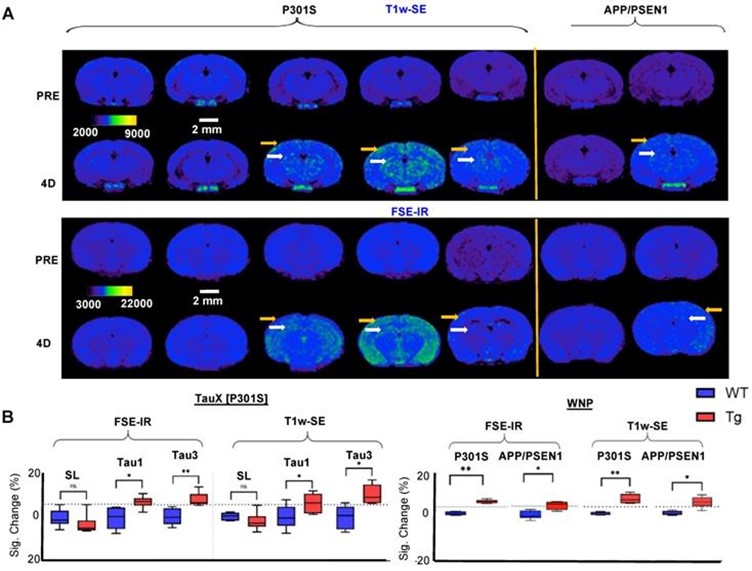
Figure 6. Magnetic resonance imaging (MRI) and immunofluorescent staining demonstrate signal enhancement as TauX binds to hyperphosphorylated tau in vivo. (A) Two-month-old mice were intravenously injected with TauX or WNPs (0.15 mmol Gd/kg). Pre- and post-contrast images for T1-weighted spin echo (T1w-SE) and fast spin echo inversion recovery (FSE-IR) demonstrate signal enhancement in delayed post-contrast scans of transgenic (TG) P301S mice treated with TauX or WNP, and APP/PSEN1 mice treated with WNP, relative to age-matched wild type (WT) controls. TG animals showed high enhancement in cortical (yellow arrow) and hippocampal regions (white arrow). TG animals showed no signal enhancement four days after injection of untargeted contrast (SL). The scale bar represents 2 mm. All animals are shown on the same color scale. (B) Box and whisker plots demonstrate signal enhancement in TG animals relative to WT counterparts and SL-treated TG animals for both T1w-SE and FSE-IR sequences (*p<0.05; **p<0.005). The dotted line indicates the signal threshold for determining sensitivity (2 standard deviations above baseline noise, ~6%).
Authors
Parag Parekh, Qingshan Mu, Andrew Badachhape, Rohan Bhavane, Mayank Srivastava, Laxman Devkota, Xianwei Sun, Prajwal Bhandari, Jason L. Eriksen, Eric Tanifum, Ketan Ghaghada, Ananth Annapragada
Texas Children’s Hospital & Baylor College of Medicine
Theranostics 2022; 12(12):5504-5521. doi:10.7150/thno.7225