Certis Oncology Intelligence:
- The Science of Certainty
About Certis
Established in 2016, Certis Oncology Solutions is a precision oncology and translational science company. It works directly with cancer patients and their oncologists to help determine the best therapies for individuals. The Certis scientific team also partners with pharmaceutical companies to help develop the next generation of anticancer therapies.
“At Certis, we want to push the boundaries of science continually,” said Certis Chief Scientific Officer Jonathan Nakashima, Ph.D. “Developing better research models and employing the most precise, objective methods for evaluating drug response is central to our business model and our mission. We believe it’s the best way to bring greater certainty to cancer patients’ treatment decisions and to mitigate drug development risk.”
Certis’s approach to studying drug efficacy is rooted in a technology considered the gold standard in cancer research: orthotopic patient-derived xenograft (O-PDX) models.1,2,3,4 O-PDX models are developed by engrafting human tumor tissue in the corresponding anatomical location in immunocompromised mice (for example, a specimen of human pancreatic cancer is implanted into the pancreas of the mouse). Because tumors are internal to the animal, they usually cannot be measured by calipers. Certis overcame this challenge and brought a whole new level of precision to tumor volume measurement with Magnetic Resonance Imaging (MRI). Certis employs the Aspect M3TM Compact MRI to generate high-resolution 3D anatomical images to monitor anticancer therapies in real-time.
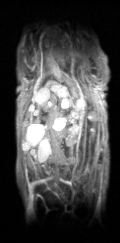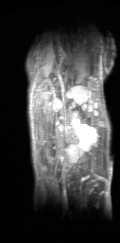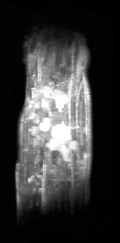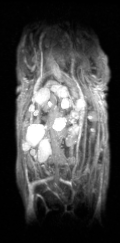
3-dimensional image shows multiple metastatic lesions of lung adenocarcinoma in the liver. Unlike tumors in subcutaneous PDX models, cancer has been shown to metastasize in O-PDX models—one of the many important advantages unique to orthotopic models
True Functional Assays of Drug Efficacy
While developing O-PDX models requires surgical, and animal husbandry expertise, the depth and quality of information derived from translational studies are unparalleled. Certis delivers true functional assays of drug efficacy on specific cancer phenotypes and subtypes and visual proof of results.

Featured Researcher:
Jonathan Nakashima, Ph.D
Chief Scientific Officer, Certis Oncology Solutions
Jonathan Nakashima oversees the company’s R&D initiatives, patient-directed orthotopic PDX (O-PDX) pharmacology studies, and the development and implementation of translational oncology assays for drug development. Dr. Nakashima has more than 15 years of experience developing preclinical oncology models. Under a California Institute for Regenerative Medicine fellowship, he received his doctorate in Medical and Molecular Pharmacology at UCLA, where he developed genetically engineered and patient-derived xenograft models to study cancers of the central and peripheral nervous systems. He continued with his post-doctoral training in the Department of Neurobiology. He received a Kirschstein National Research Service Award to study the cell-interactions in the brain tumor microenvironment.
References
- Russell T.A., Eckardt M.D., Murakami T., Singh A.S., Kiyuna T., Igarashi K., Kawaguchi K., Li Y., Crompton J., Dry S.M., Federman N., Chmielowski B., Shurrell E., Hoffman R.M., Eilber F.C. Clinical factors that affect the establishment of soft tissue sarcoma patient-derived orthotopic xenografts: A UCLA sarcoma program prospective clinical trial. JCO Precision Oncology, 2017. | READ
- Guerin MV, Finisguerra V, Van den Eynde BJ, Bercovici N, Trautmann A. Preclinical murine tumor models: a structural and functional perspective. Elife. 2020 Jan 28;9:e50740. doi: 10.7554/eLife.50740. PMID: 31990272; PMCID: PMC6986875. READ
- Devaud C, Westwood JA, John LB, Flynn JK, Paquet-Fifield S, Duong CP, Yong CS, Pegram HJ, Stacker SA, Achen MG, Stewart TJ, Snyder LA, Teng MW, Smyth MJ, Darcy PK, Kershaw MH. Tissues in different anatomical sites can sculpt and vary the tumor microenvironment to affect responses to therapy. Mol Ther. 2014 Jan;22(1):18-27. doi: 10.1038/mt.2013.219. Epub 2013 Sep 19. PMID: 24048441; PMCID: PMC3978809. READ
- Kamili, A., Gifford, A.J., Li, N. et al.Accelerating development of high-risk neuroblastoma patient-derived xenograft models for preclinical testing and personalised therapy. Br J Cancer 122, 680–691 (2020). READ