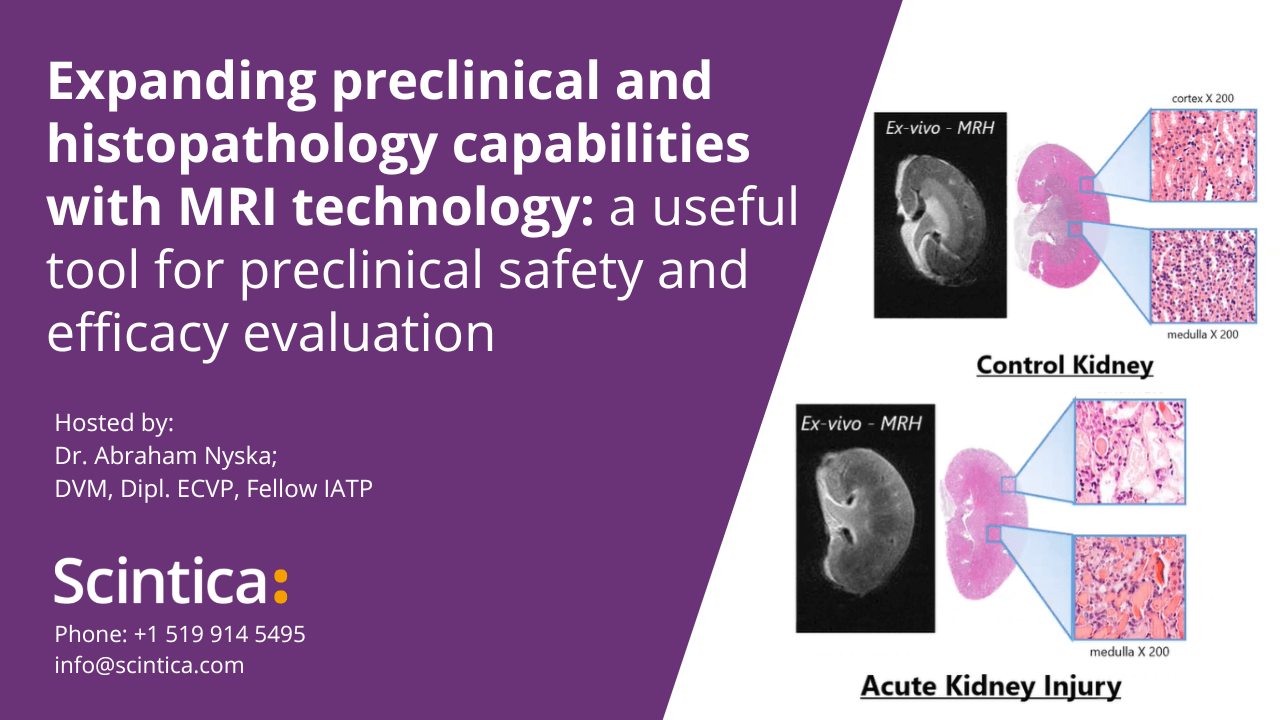
[WEBINAR] Expanding preclinical and histopathology capabilities with MRI technology: a useful tool for preclinical safety and efficacy evaluation
This free webinar hosted by Scintica Instrumentation reviewed the fundamentals of Magnetic Resonance Histology (MRH) and provided a number of relevant examples. Magnetic Resonance Imaging (MRI) has been used for years in preclinical research to perform in vivo studies allowing for the sensitive detection of pathological changes in soft tissue and to provide quantitative three-dimensional data.
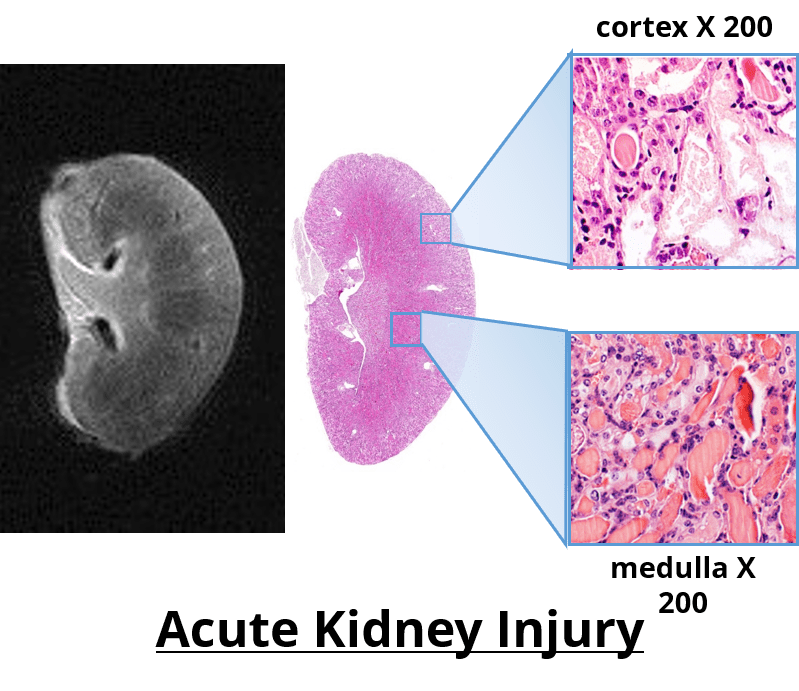
It has been used in longitudinal studies to noninvasively monitor the genesis, progression and regression of a wide variety of diseases, reducing the need for interim sacrifice of animals at specified time points, thus allowing the same animal to be used as its own control within a given study. MRH is the use of MR imaging on formalin-fixed tissues for high resolution characterization of tissue structure. It is a highly valuable complimentary adjunct to conventional histopathology, as it permits a thorough examination to be performed through multiple digital slices of an entire organ, while leaving the formalin-fixed specimen intact for subsequent definitive conventional diagnostic histopathology.
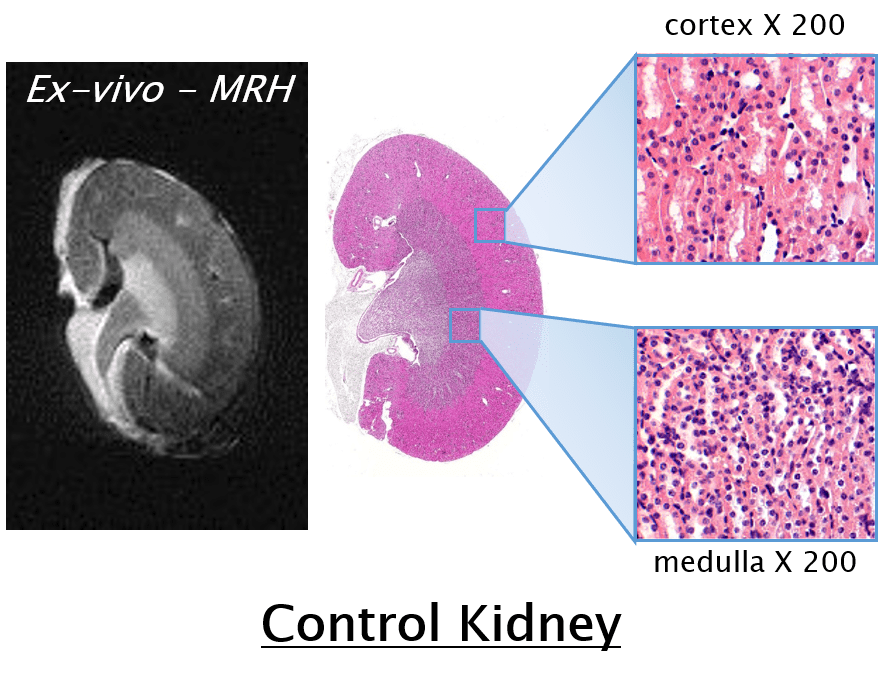
Specifically, as it relates to safety evaluations and toxicology studies the Federal Drug Administration (FDA) has followed suite with the Society of Toxicologic Pathology’s (STP) recommendation to increase the required number of samples taken for conventional histology to enhance the sensitivity of detecting toxic effects. However, the consensus still remains that that this increased sampling does not suffice to cover the entire organ, and that some lesions may be missed. If however MRH was used to characterize the entire organ of interest, these 3D images could then be used to accurately identify effected regions, which would then be sampled for confirmation by conventional histopathology.
In addition to safety pharmacology and toxicology MRH has wide applicability when examining the changes caused by implantation of medical devices, injection/infusion site inflammation following administration of a target compound, therapeutic stem cell injections which may cause teratology over time, and of course in studying a wide variety of cancers and fibrotic diseases. In all cases the ideal imaging protocol would be to longitudinal follow the in vivo study non-invasively, upon termination of the studies the tissues could then be removed and fixed in formalin. MRH would then be used to digitally examine the entire organ in 3D, with the results used to guide the sectioning of the specimen for conventional histopathological analysis and characterization of the lesions.
 This webinar demonstrated how compact MRI technology can serve as an important adjunct to toxicology and toxicologic pathology by non-destructively providing 3D imaging data, detailed morphological insights, and quantitative information. Specifically this webinar discussed the following:
This webinar demonstrated how compact MRI technology can serve as an important adjunct to toxicology and toxicologic pathology by non-destructively providing 3D imaging data, detailed morphological insights, and quantitative information. Specifically this webinar discussed the following:
- Fundamentals of Magnetic Resonance Histology (MRH)
- Practical examples relevant to:
- Carcinogenicity testing
- Safety (toxicity) evaluation
- Injection/infusion site inflammation following compound administration
- Cancer biology
- Pulmonary fibrosis
Relevant Publications by Dr. Abraham Nyska:
About the Speaker (s)

Dr. Abraham Nyska
DVM, Dipl. ECVP, Fellow IATP
Dr. Abraham Nyska is an expert in toxicological pathology with more than 40 years of experience in the preclinical pathological evaluation and risk assessment of drugs, chemicals, medical devices, and stem cells. He worked for 18 years as a staff scientist and consultant at the American National Toxicology Program of the National Institute of Health, and continues working as a consultant in toxicologic pathology to a variety of pharmaceutical companies, contract research organizations, and federal institutes in Israel, Europe, India, and North America. He is currently a visiting full professor of pathology at Tel Aviv University in Israel, and is a co-author on more than 440 peer reviewed publications and/or book chapters.
Dr. Nyska’s vision is to provide a professional service to researchers in the environmental sciences and related disciplines by advancing knowledge to enhance human health. His passion lies in disseminating the importance of MRH for toxicology and toxicologic pathology applications, as well as in the application of state-of-the-art digital telepathology solutions.