Preclinical Imaging of Conscious Animals
System Used:
SuperArgus

Introduction
Molecular imaging techniques such as positron emission tomography (PET), magnetic resonance imaging (MRI), intravital microscopy (IVM), and single photon emission computed tomography (SPECT) are valuable tools for studying brain function in preclinical animal models. To accomplish this, animals are typically anesthetized to reduce unwanted artifacts that can be caused by animal movement. Unfortunately, anesthesia has been shown to have detrimental effects on developing mouse tissues, particularly the brain, suggesting the assessment of animal functions while under anesthesia could lead to inaccurate findings.
To circumvent these concerns, several imaging strategies have been developed to enable non-invasive assessment of brain function in conscious awake animals without the effect of anesthesia. These techniques provide an opportunity to study important concepts within cognitive and behavioral neuroscience that will likely lead to more accurate disease modeling and improved animal welfare for subjects of preclinical research.
In a recent paper published in Scientific Reports, Karda et al. performed bioluminescence imaging (BLI) in conscious, unrestrained mice following the administration of lentiviral cassettes (biosensors) encoding luciferase. For the first time, they demonstrate that whole body bioluminescence imaging can be performed on conscious mice throughout the entire lifespan, using organ-specific biosensors1. Similarly, a team at Johns Hopkins University developed the first SPECT system in 2013 to perform imaging of conscious, unrestrained animals. To do this, they tracked the head position of an awake animal using NIR cameras and then corrected it for motion during the image reconstruction process. With this approach, they were able to quantify the binding potential of the transporter radioligand 123I-isoflurane which is often used to evaluate several clinical neurodegenerative diseases2
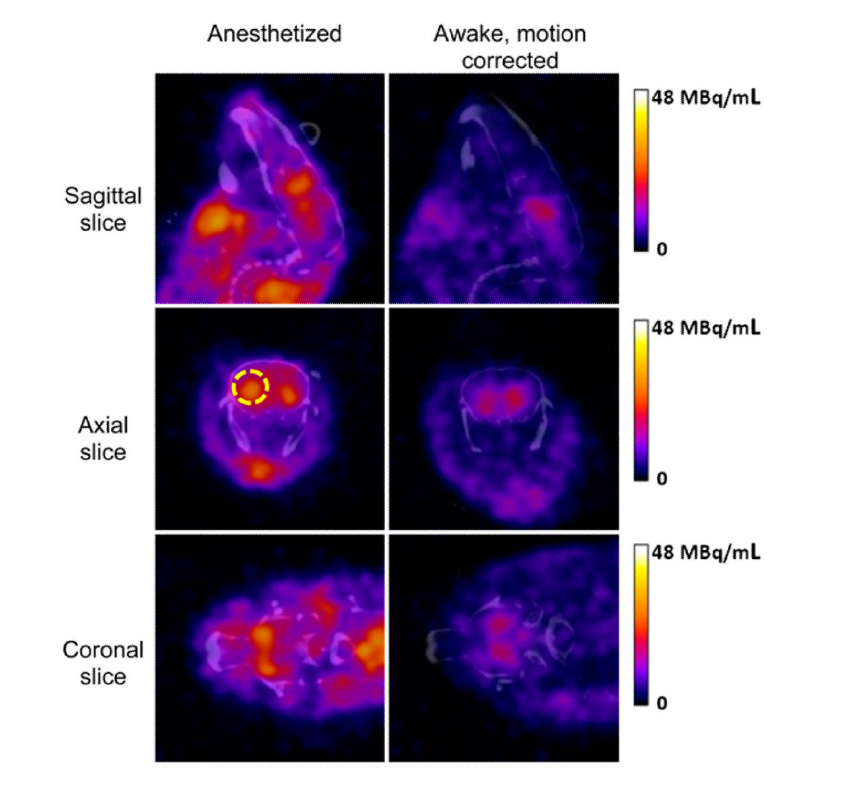
Figure 1: Image reconstruction results for 123I-ioflupane SPECT scans of the anesthetized mouse (left) and awake mouse (right) co-registered with CT. Higher striatal uptake was observed in anesthetized mice. Image scale peak of 48 MBq/mL corresponded to approximately 29% of injected dose/mL. Right striatum was identified on transaxial images (yellow broken circle)2.
In the last decade, many have tried to explore the use of PET imaging for studying brain function in awake animals including the optimization of head restraining techniques and the use of motion-tracking devices. One such approach has been made possible by Sedecal, the manufacturer of the SuperArgus preclinical PET/CT system. This system is equipped with unique phoswitch detectors which provide true depth of interaction (DOI) information and ultimately lead to improved sensitivity for real-time PET imaging.
How Does it Work?
Prior to the imaging session, the animal is conditioned to an imaging tube, made of translucent red plastic so that the animal remains calm but can still be observed. The diameter of the tube is selected such that the animal can’t move around freely but still has sufficient room to prevent being compressed. In the case of neurological imaging, fiducial markers are placed on either side of the nose and behind each ear. Once positioned within the tube, the tracer is administered either before or during the imaging session depending on the research question, and images of the awake animal are acquired.
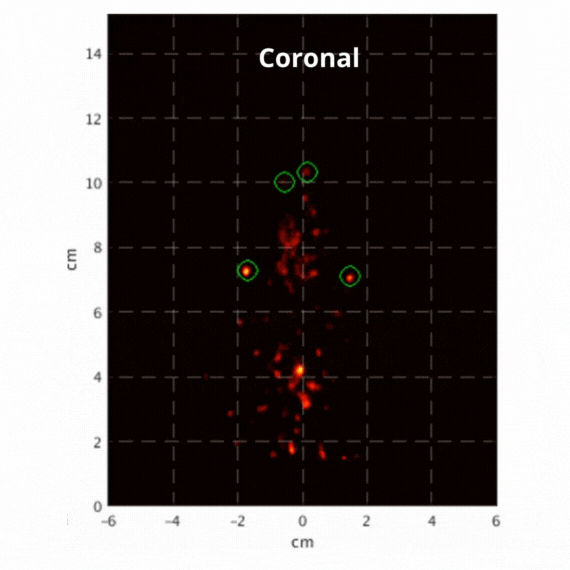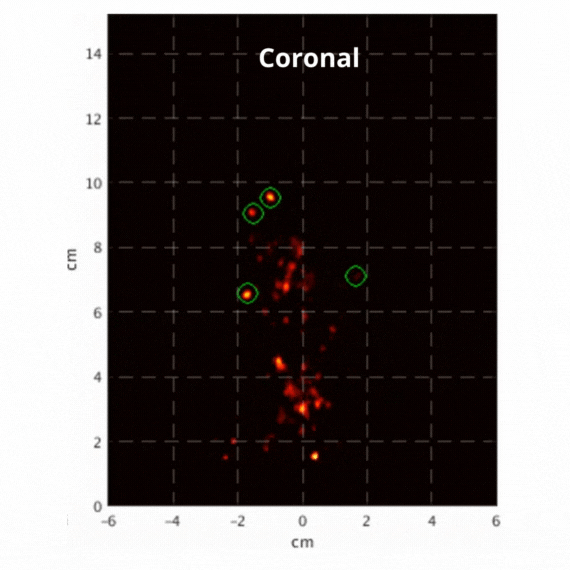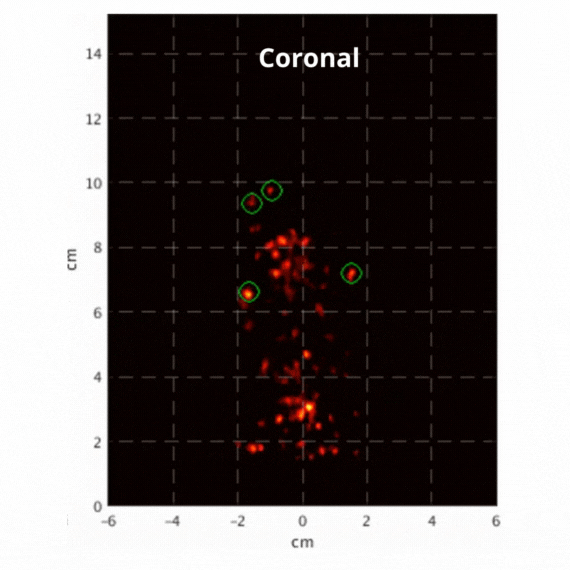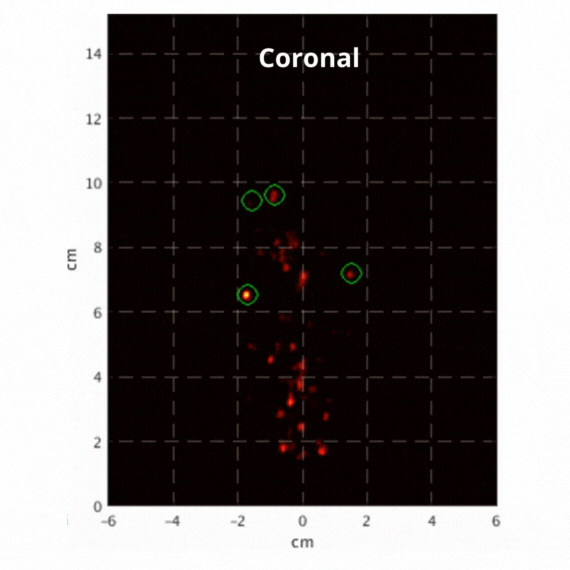
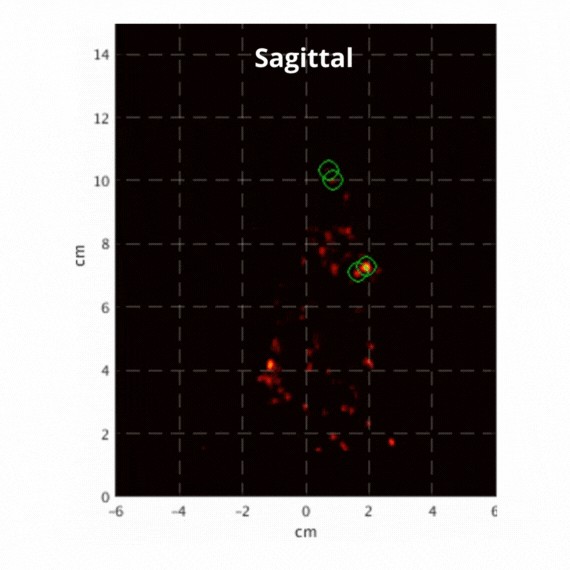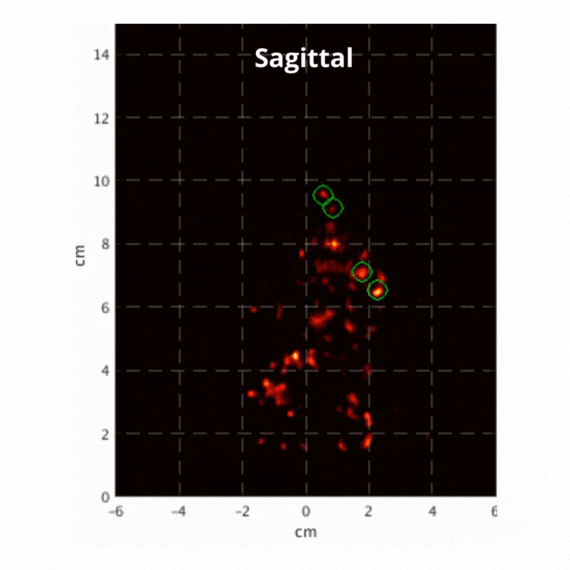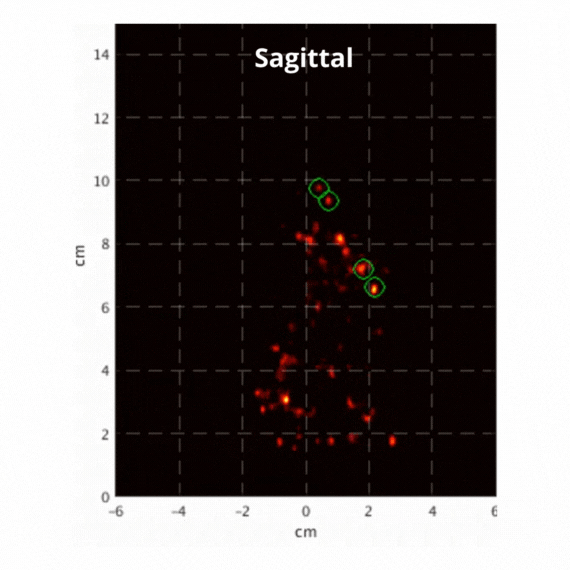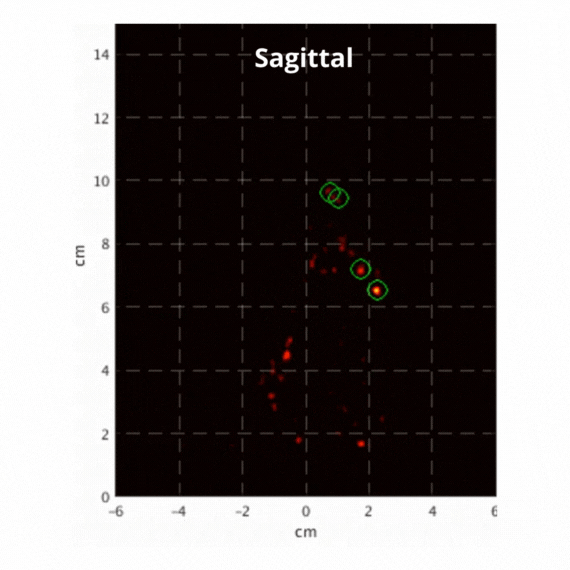
Figure 2: Resulting images of an awake rat using the SuperArgus PET/CT system reconstructed at 50 frames per second. Fiducial markers are circled in green, and the resulting signal is seen throughout the brain in red. The movement from the fiducial markers and signal is seen as the animal is awake and under no form of sedation. Sagittal view (L) and coronal view (R).
Future Outlook
Having the ability to study unanesthetized animals with quantitative imaging technologies may greatly improve our understanding of many different neurochemical processes and their effects on brain function and metabolism. For example, these tools could be used to quantify complex pharmacological responses such as the rate of synthesis, transport, and receptor expression, or to study neuronal activation in response to stimulation. These techniques have not been widely adapted yet due to the additional hardware and/or complicated software that is often required. However, as we gain more insight into how anesthesia can affect normal development and biological processes, it will become crucial that these novel imaging strategies are optimized, streamlined, and adapted for in vivo neurological research.
Authors
Tyle Lalonde Ph.D. – Product Manager – Nuclear Medicine – Scintica
Katie Parkins Ph.D. – Head of Scientific Integration – Scintica
References
1. Karda, R. et al. Continual conscious bioluminescent imaging in freely moving somatotransgenic mice. Sci Rep 7, (2017).
2. Baba, J. S. et al. Molecular imaging of conscious, unrestrained mice with AwakeSPECT. Journal of Nuclear Medicine 54, 969–976 (2013).