Exosome-based delivery of super-repressor IκBα ameliorates kidney ischemia-reperfusion injury
Seonghun Kim1,7, Sul A Lee2,3,7, Heakyung Yoon4, Myung Yoon Kim4, Jae-Kwang Yoo4, So-Hee Ahn4, Cheol Hyoung Park4, Jimin Park5, Bo Young Nam5, Jung Tak Park2, Seung Hyeok Han2, Shin-Wook Kang2, Nam Hee Kim1, Hyun Sil Kim1 , Dawool Han1, Jong In Yook1, Chulhee Choi4,6 and Tae-Hyun Yoo2
System Used:
IVM

Abstract
Ischemia-reperfusion injury is a major cause of acute kidney injury. Recent studies on the pathophysiology of ischemia-reperfusion-induced acute kidney injury showed that immunologic responses significantly affect kidney ischemia-reperfusion injury and repair. Nuclear factor (NF)- kB signaling, which controls cytokine production and cell survival, is significantly involved in ischemia-reperfusioninduced acute kidney injury, and its inhibition can ameliorate ischemic acute kidney injury.
Using EXPLOR, a novel, optogenetically engineered exosome technology, we successfully delivered the exosomal super-repressor inhibitor of NF-kB (Exo-srIkB) into B6 wild type mice before/ after kidney ischemia-reperfusion surgery, and compared outcomes with those of a control exosome (Exo-Naïve)- injected group. Exo-srIkB treatment resulted in lower levels of serum blood urea nitrogen, creatinine, and neutrophil gelatinase-associated lipocalin in post-ischemic mice than in the Exo-Naïve treatment group. Systemic delivery of ExosrIkB decreased NF-kB activity in post-ischemic kidneys and reduced apoptosis.
Post-ischemic kidneys showed decreased gene expression of pro-inflammatory cytokines and adhesion molecules with Exo-srIkB treatment as compared with the control. Intravital imaging confirmed the uptake of exosomes in neutrophils and macrophages. Exo-srIkB treatment also significantly affected postischemic kidney immune cell populations, lowering neutrophil, monocyte/macrophage, and T cell frequencies than those in the control. Thus, modulation of NF-kB signaling through exosomal delivery can be used as a novel
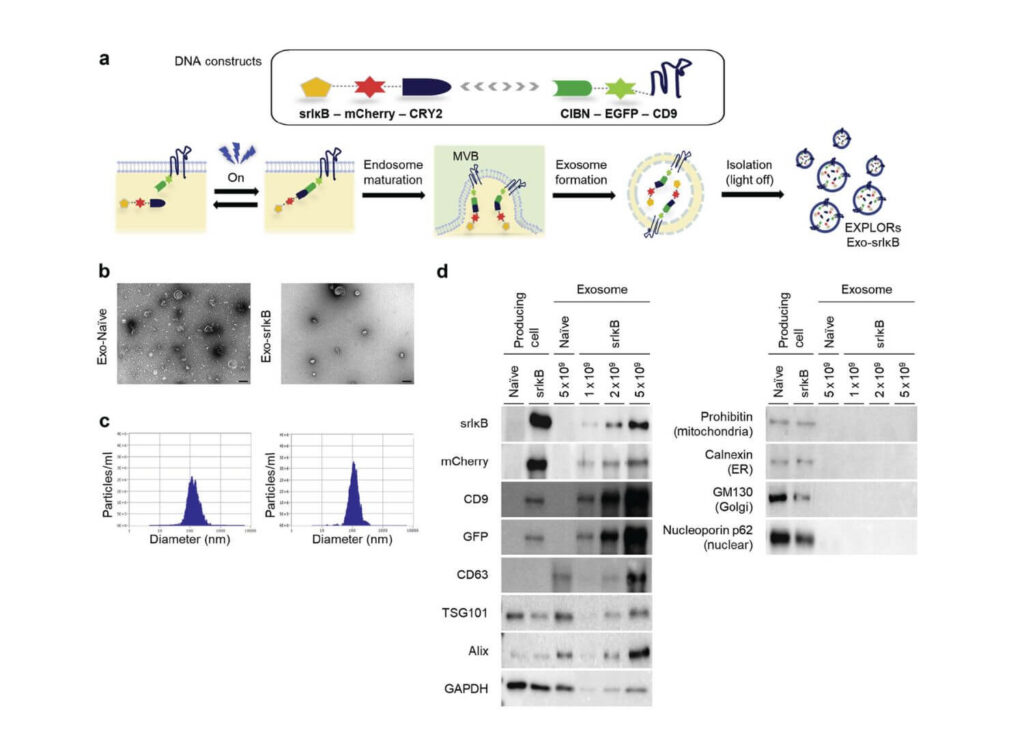
Figure 1 | Characterization of engineered exosomes produced from human embryonic kidney (HEK)293T. (a) Schematic diagram of DNA constructs used for producing super-repressor kappaB inhibitor (IkB)–loaded exosomes (Exo-srIkB) (upper), and biogenesis of cargo protein–carrying exosomes using optically reversible protein–protein interactions, so-called exosomes for protein loading via optically reversible protein-protein interactions (EXPLOR) technology (lower). (b) Representative transmission electron microscopy images of a control exosome (Exo-Naïve) and Exo-srIkB. Bar ¼ 100 nm. (c) Concentrations and size distributions of Exo-Naïve (left) and Exo-srIkB (right) were determined by a Zetaview instrument (Particle Metrix, Germany). (d) Immunoblotting HEK293T cells and HEK293T cell–derived exosomes to analyze the expression of target proteins (srIkB, mCherry, CD9, and green fluorescent protein [GFP]), exosome-positive markers (CD63, TSG101, Alix, and glyceraldehyde-3-phosphate dehydrogenase [GAPDH]), and exosome-negative markers (cell organelle markers; prohibitin, calnexin, GM130, and nucleoporin p62). Naïve cells and Exo-Naïve cells were used as negative controls of Exo-srIkB. CIBN, N-terminal fragment of cryptochrom-interacting basic-helix-loop-helix 1; EGFP, enhanced green fluorescent protein.