System Used:
SuperArgus

 Abdominal aortic aneurysm (AAA) is a localized dilatation of the abdominal aorta which shows limited symptomology prior to rupture. Once rupture occurs though, the results may be catastrophic leading to intense pain, loss of consciousness, and possibly death. Detection often occurs incidentally or through routine screening, and surgical intervention is generally limited to large aneurysms (> 5.5 cm). If detected below this limit, surgical intervention is avoided, and patients are required to undergo frequent ultrasound screening, sometimes for many years, to monitor aneurysm progression. This places a considerable burden on the healthcare system and the patients, many of whom are over the age of 50. A non-invasive biomarker which tracks cellular proliferation within the aneurysm may provide the necessary information to stratify patients through personalized monitoring and treatment approaches, thus reducing the strain of repeated screening visits.
In this paper from 2019 published in the Journal of Nuclear Cardiology, researchers demonstrated the feasibility of fluorine-18-fluorothymidine ([18F]FLT) as a cell proliferation radiotracer for PET/ CT in the progressive angiotensin II (AngII) infusion pre-clinical model of AAA. In this model, apolipoprotein E deficient (apoE−/−) mice receive infusions of human AngII via osmotic minipumps which leads to injuries that mimic many facets of the human presentation of AAA and follow a well-defined time course of events.
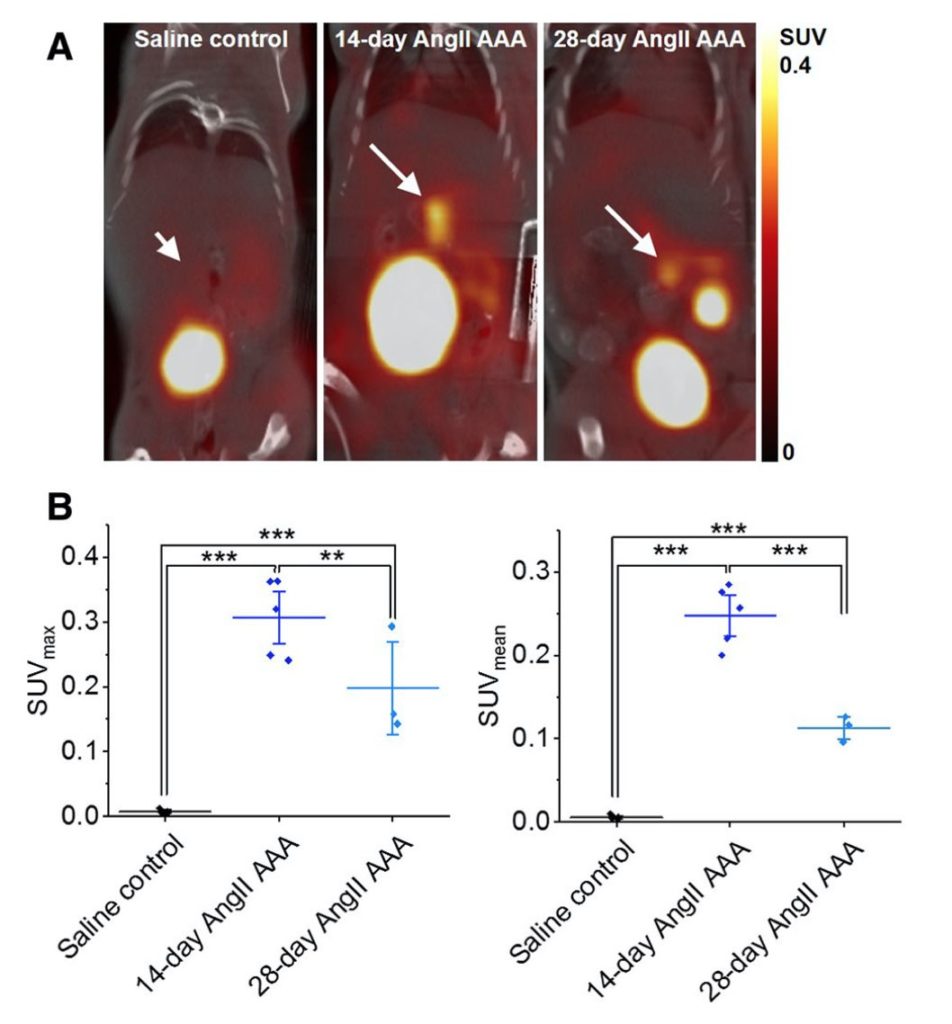
In this study, mice underwent 90-minute dynamic [18F]FLT PET/CT after 14 and 28 days of AngII infusion using the Sedecal SuperArgus PET/CT. [[18F]FLT uptake was significantly greater in 14-day and 28-day AAA than in control aortae. Ex-vivo gamma counting and three dimensional ultrasound confirmed a positive correlation between [18F]FLT uptake and aortic volume. These results show the feasibility of [18F] FLT as a PET/CT radiotracer and further suggest the possibility of this radiotracer to monitor progression and predict AAA rupture.
For more information about the Sedecal SuperArgus PET/CT system or our preclinical ultrasound systems, please contact Scintica Instrumentation (info@scintica.com).
Abdominal aortic aneurysm (AAA) is a localized dilatation of the abdominal aorta which shows limited symptomology prior to rupture. Once rupture occurs though, the results may be catastrophic leading to intense pain, loss of consciousness, and possibly death. Detection often occurs incidentally or through routine screening, and surgical intervention is generally limited to large aneurysms (> 5.5 cm). If detected below this limit, surgical intervention is avoided, and patients are required to undergo frequent ultrasound screening, sometimes for many years, to monitor aneurysm progression. This places a considerable burden on the healthcare system and the patients, many of whom are over the age of 50. A non-invasive biomarker which tracks cellular proliferation within the aneurysm may provide the necessary information to stratify patients through personalized monitoring and treatment approaches, thus reducing the strain of repeated screening visits.
In this paper from 2019 published in the Journal of Nuclear Cardiology, researchers demonstrated the feasibility of fluorine-18-fluorothymidine ([18F]FLT) as a cell proliferation radiotracer for PET/ CT in the progressive angiotensin II (AngII) infusion pre-clinical model of AAA. In this model, apolipoprotein E deficient (apoE−/−) mice receive infusions of human AngII via osmotic minipumps which leads to injuries that mimic many facets of the human presentation of AAA and follow a well-defined time course of events.
In this study, mice underwent 90-minute dynamic [18F]FLT PET/CT after 14 and 28 days of AngII infusion using the Sedecal SuperArgus PET/CT. [[18F]FLT uptake was significantly greater in 14-day and 28-day AAA than in control aortae. Ex-vivo gamma counting and three dimensional ultrasound confirmed a positive correlation between [18F]FLT uptake and aortic volume. These results show the feasibility of [18F] FLT as a PET/CT radiotracer and further suggest the possibility of this radiotracer to monitor progression and predict AAA rupture.
For more information about the Sedecal SuperArgus PET/CT system or our preclinical ultrasound systems, please contact Scintica Instrumentation (info@scintica.com).