Cancer Biology and Magnetic Resonance Imaging (MRI): The Gold Standard of Soft Tissue Imaging
System Used:
M-Series MRI

Magnetic resonance imaging (MRI) is a technique that utilizes magnetic fields to create images of the anatomy and the physiological processes within the body. Specifically, MRI, the gold standard in soft tissue imaging, offers researchers the ability to distinctly contrast between normal and pathological tissue.
When it comes to the strengths of MRI in cancer biology, MR allows for better longitudinal exploration of the tumor microenvironment in both human patients and animal models. Exploring the differences between normal adjacent tissue and the tumor microenvironment surrounding tumor tissue, allows the focus of cancer biology to shift from solving individual questions of tumorgenicity to understanding tumor progression from a whole systems point of view. Better still, since the MRI is based on a magnetic field, rather than radioactive labeling, MRI provides a safer option for longitudinal study than computed tomography (CT) or positron emission tomography (PET).
In this article, we are going to explore current applications of MR Imaging in preclinical cancer research. These applications include:
- Tumor visualization and quantification
- Contrast agent imaging
- Multi-modal tumor imaging
- MR-Based Histology
Tumor Visualization and Quantification
When we think about MR Imaging in cancer biology, tumor visualization and quantification is the easiest application to consider. MR Imaging is considered the gold standard in soft tissue imaging because of its ability to produce the clearest image of what is happening within the soft tissue of the body. In tumor research, being able to visualize and quantify soft tissue tumors is of vital importance, not just in the staging and grading of tumors, but in defining the successes of treatments or the successes, unfortunately, of tumor-inducing agents. In a clinical setting, MR Imaging is relied upon for an accurate visualization of tumor and aggression, as well as a longitudinal study in efficacy of treatment.
Until the recent availability of affordable, easy to use, and compact MR systems, from companies like Aspect Imaging, the same longitudinal studies have not been as readily accessible in preclinical research. This has left a huge gap in our understanding of cancer progression and treatment efficacy and will hopefully change soon.
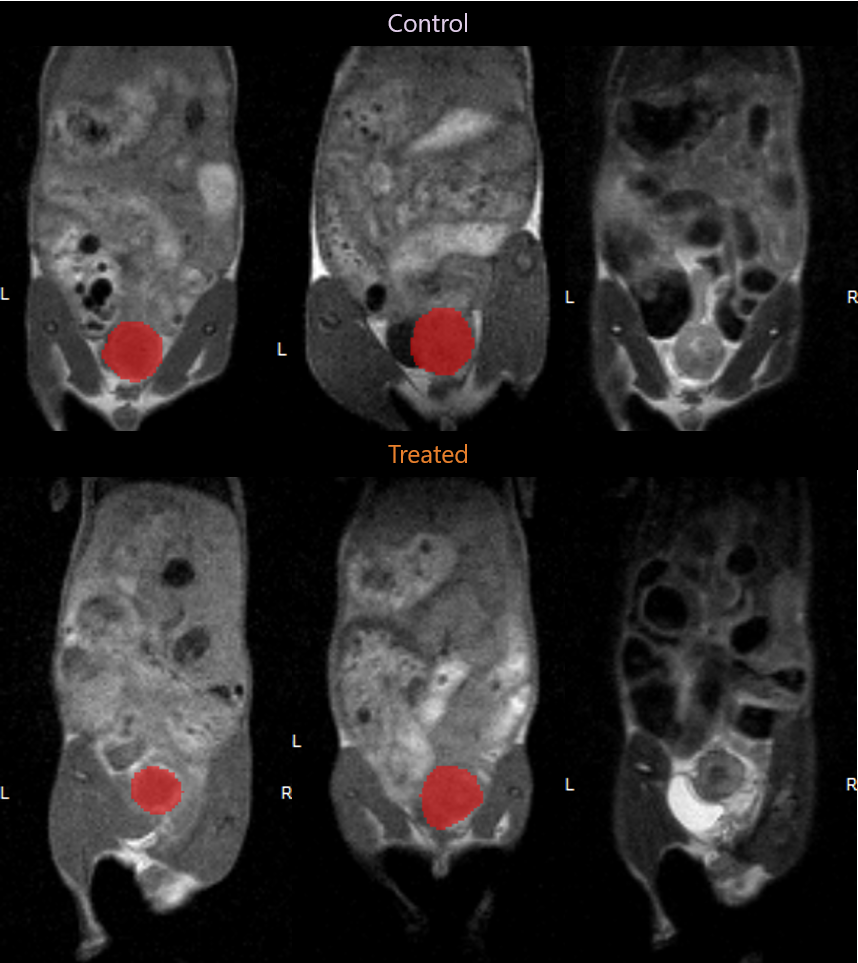
Tumor Volume calculation made possible by MR imaging.
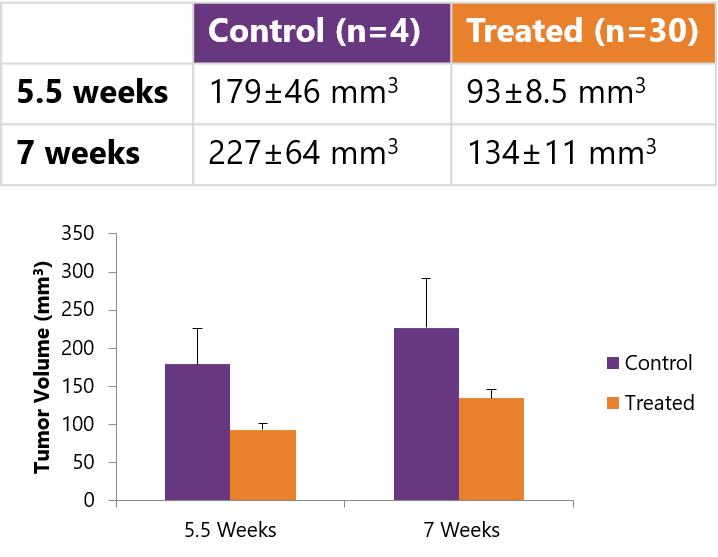
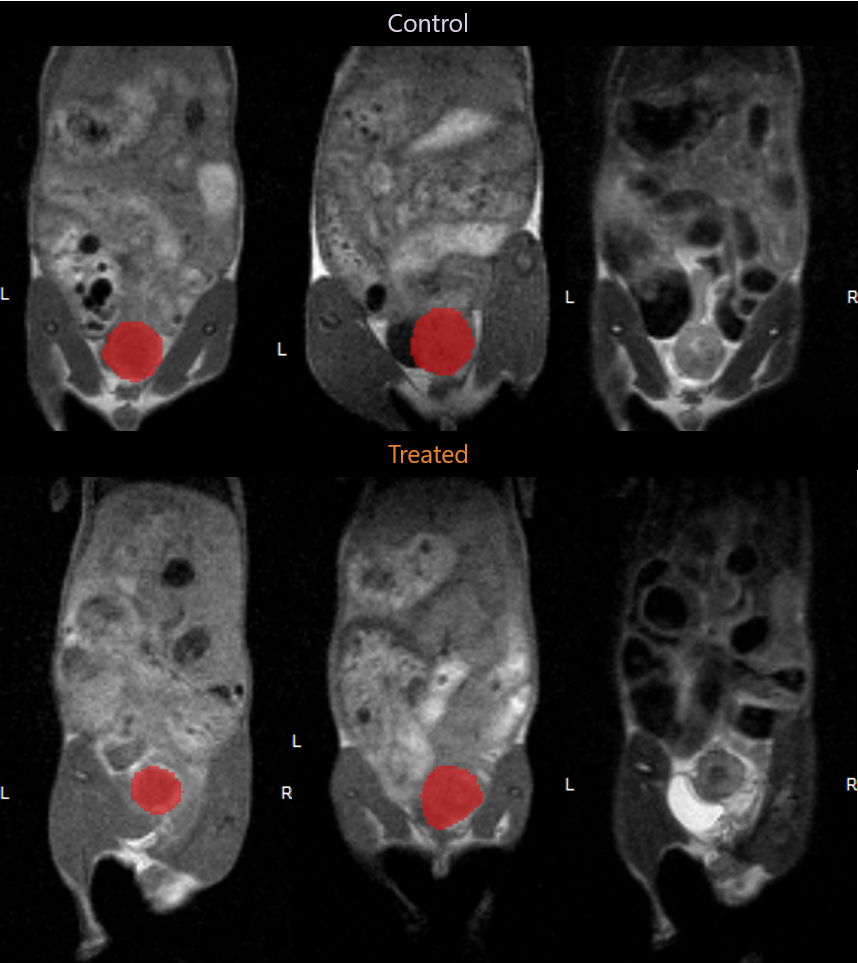
Tumor Volume calculation made possible by MR imaging.
Contrast Agent Imaging
With the ever expanding library of contrast agents in MR Imaging, the ability to study perfusion, angiogenesis, and specific molecular targets is an often underexplored option for MR Imaging in cancer research. By utilizing gadolinium or iron oxide-based contrast agents researchers can more easily identify, define, and quantify tumor tissues based on perfusion or molecular characteristics. Further, with the option to customize these contrast agents with specific antibodies, MR-based histology becomes a limitless area of cancer exploration.
Multi-Modal Tumor Imaging
With the adaptation of most clinically relevant imaging modalities to preclinical research models, the ability to harness information from a multitude of imaging modalities allows researchers to gather more relevant information about the tumor and surrounding microenvironment than ever before. Co-registration of these images, taken simultaneously or sequentially, provides invaluable insights to further the understanding over the course of a longitudinal study.
Examples include PET/MRI, or even 3D optical images co-registered with a whole body MRI image. The molecular information provided by the PET or optical images (both of which have their own unique capabilities and strengths) help the researcher identify the general tumor location or specific target of interest, while the co-registered MRI image can be used to provide the anatomical context, soft tissue differentiation, and image resolution to be able to accurately confirm the presence of the tumor and provide a means to quantify the lesions size. Combined, at specified time-points within a study, these insights may be necessary to more fully elucidate the pathological effects or therapeutic response occurring within a specific animal model.

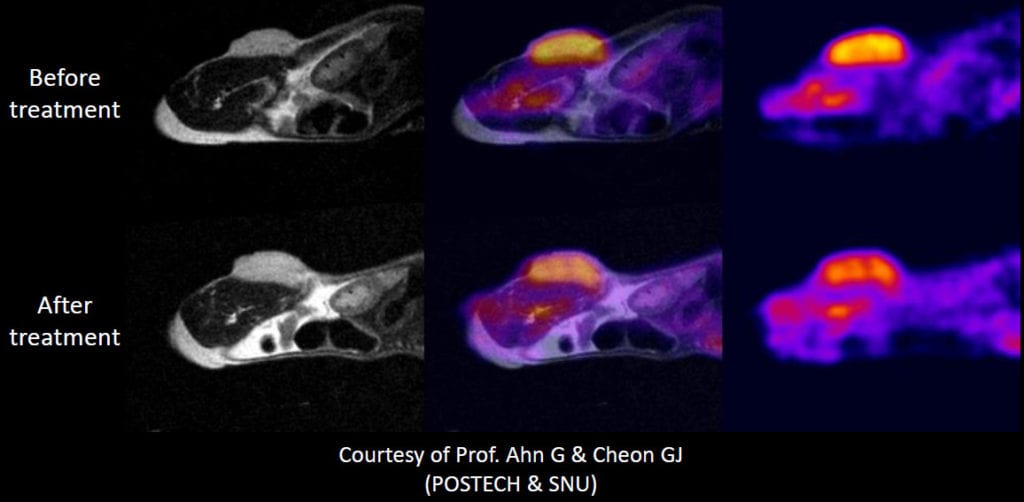
Co-registration of PET and MRI images yield more information than either would alone.
MR-Based Histology
MR Histology (MRH) typically refers to the ex vivo imaging of a fixed tissue sample to provide very high resolution images of the whole organ prior to proceeding to conventional histological sectioning and staining.
In some cases an organ may have very few lesions present and it can be invaluable to predetermine the number, location, and size of these lesions for a more complete understanding of the pathology. Also, this insight can be used to help guide the sectioning of the organ for histological analysis, in this way ensuring that appropriate slices are taken without the need for sectioning and analyzing the entire organ.
Conventional histological and immunohistochemical staining can then be completed on the samples to confirm the presence of the lesion, identify the pathology more definitively, and further explore any specific molecular targets of interest.
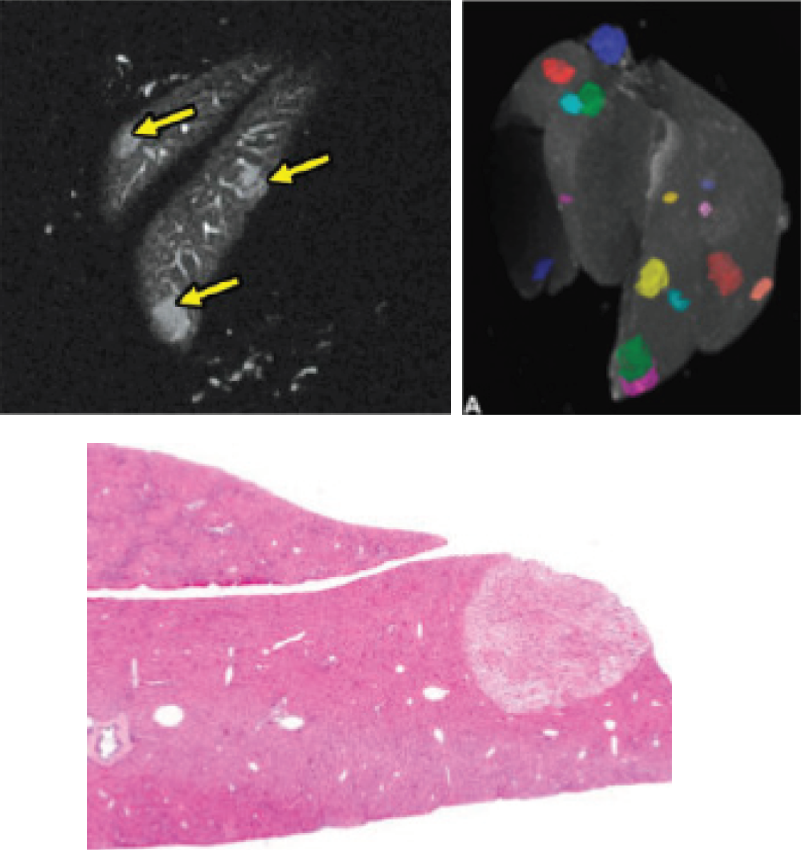
Comparison of histological sample and MR-based histology image.
Cost Effective Preclinical MR Imaging Solutions
The M-Series Compact MRI system offers cancer researchers an affordable, easily accessible method for defining and quantifying tumor tissue, and comparing pathological lesions to adjacent normal tissue. Operating at 1 Tesla, this system provides enhanced sensitivity to Gadolinium based contrast agents, and performs well when using iron oxide nanoparticles. With specific add-ons, this system can provide simultaneous PET/MR images, or sequential MRI and optical (designed to work with third part optical systems) images, as well as automated MR histology sample handing. Further, access to an affordable MRI system in preclinical research enhances the quality of research and information being derived from one’s experiments without the need to allocate funds to a separate imaging core for expertise or usage.
For more information on this system, please visit the M-Series Compact MRI system page or feel free to contact us at +1 519 914 5495.