System Used:
M-Series MRI

Glioblastomas (GBMs) are the most common malignant primary tumor of the central nervous system, accounting for over half of all gliomas1. With the current gold standard of treatment being surgical resection, magnetic resonance imaging (MRI) continues to play a huge role in defining tumor margins and evaluating treatment response in the clinic. However, while it is considered the superior modality for soft tissue contrast, MRI has limited sensitivity and specificity. Similarly, fluorescence imaging is commonly used to define tumor edges for high-grade glioma surgery but photobleaching, phototoxicity and tissue autofluorescence limit its potential efficacy. Thus, a novel imaging strategy is urgently needed to better visualize and characterize GBM throughout disease progression.
The role of molecular imaging
Molecular imaging plays a vital role in diagnosing, monitoring, and treating various types of cancer. Previous studies have shown that multimodal imaging can be used in certain applications to overcome the limitations of using each modality independently2. For example, modalities that are conventionally used for anatomical imaging such as MRI or CT can be very complementary to functional imaging technologies, including optical and PET. Furthermore, multimodal reporter genes are being developed that can be visualized using more than one technology. This will provide opportunity for additional information to be acquired as well as offer flexibility for sites that have limited access to imaging equipment.
In a recent study, Huang et al. developed a targeted nanoparticle that enables trimodal imaging as a diagnostic and therapeutic strategy for GBM. The molecular probe which targeted oncogenes EGF and SEC61G, demonstrated T2 contrast on MRI and well-defined boundaries using photoacoustic imaging. Furthermore, using near-infrared fluorescence imaging they show the probe has high specificity to tumor cells and prolonged durability in vivo. Overall, this biotarget strategy shows great promise for a comprehensive and translationally relevant imaging platform for clinical cases of GBM3.
In this study, longitudinal MRI (M-Series from Aspect Imaging), and bioluminescence imaging (BLI) (Newton 7.0 from Vilber) were used to monitor tumor growth every three days. MRI and BLI are often used in combination for visualizing and quantifying various biomarkers in vivo. For tumor studies, they provide very complementary information. MRI provides detailed information on the size and location of the tumor, while fluorescence BLI provides accurate measurements of cell viability throughout the tumor. This can be especially important for treatment studies as the size of the tumor may not change significantly over time however, the amount of viable tissue may change quite quickly as the result of an effective treatment.
Additionally, Huang et al. used photoacoustic imaging to provide high sensitivity images of the tumor boundaries. They found that the tumor profile measured by PAI matched histological boundaries very well, which can be attributed to the high expression of EGFR and SEC61G on the outer edges of the tumor. As a result, PAI may provide important complementary information to MR and optical methods during surgical intervention.
This study is one of many demonstrating significant promise for targeted multimodal theranostics in the cancer field. If you’re interested in learning more about the imaging systems used throughout this study, check out the product pages on our website below.
For more information about the M-Series Compact MRI & Newton 7.0 Optical Imaging System, please contact Scintica Instrumentation (info@scintica.com).
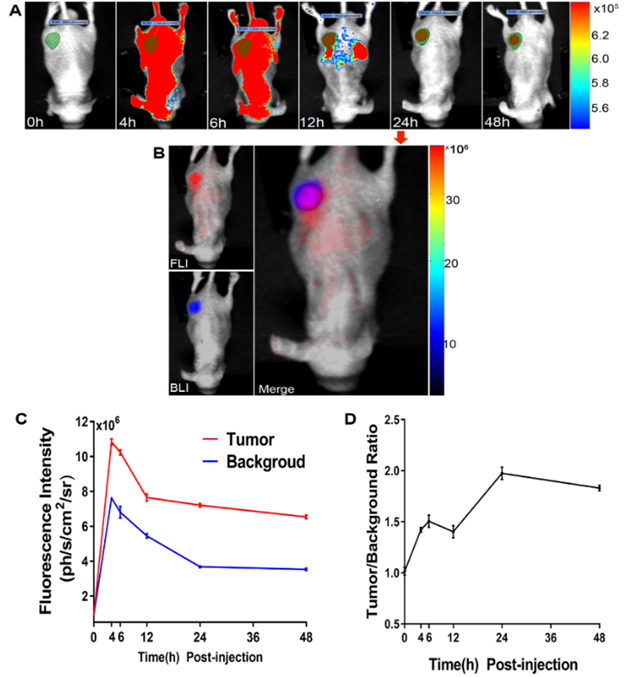
Fluorescence images of dual-targeted probes. (A) To test and verify its best imaging time point, the post-injection images of 0, 4, 6, 12, 24, and 48 h were acquired in a subcutaneous tumor model. (B) The fluorescence & bioluminescence and their merged images at 24 h after injection in vivo. (C & D) The fluorescence intensity (C) and signal-to-noise ratios (D) of different time points were recorded and calculated. (*Figure from Huang et al / Nanomedicine: Nanotechnology, Biology, and Medicine 40 (2022) 102509).

T2-weighted MR images of non/dual-targeting probes. The typical original MR images, as well as the matched T2 color map images of dual-targeting (A) and non-targeting(C) probes, were presented. (B & D) The corresponding T2 value changes over time. (*Figure from Huang et al / Nanomedicine: Nanotechnology, Biology, and Medicine 40 (2022) 102509).
References
1. American Brain Tumor Association. Retrieved February 08, 2022. https://www.abta.org/tumor_types/glioblastoma-gbm/
2. D. Haeusler, C. Decristoforo, J. Frost, S. Gobalakrishnan, Y. Y. Huang, “Molecular Imaging: In Vivo Agents for the Diagnosis and Treatment of Cancer”, Contrast Media; Molecular Imaging, vol. 2018, Article ID 8541915, 2 pages, 2018. https://doi.org/10.1155/2018/8541915
3. Qi. Huang, K. Wang, S. Wanggou, J. Tian, X. Li. “A novel co-targeting strategy of EGFR/SEC61G for multi-modality fluorescence/MR/photoacoustic imaging of glioblastoma” Nanomedicine: Nanotechnology, Biology and Medicine, Volume 40, 2022, 102509, ISSN 1549-9634, https://doi.org/10.1016/j.nano.2021.102509.